Effects of a Personalized Physical Exercise Program in the Arterial Stiffness in Older Adults
- DOI
- 10.2991/artres.k.191126.001How to use a DOI?
- Keywords
- Ageing; arterial stiffness; pulse wave velocity; exercise
- Abstract
Introduction: This study was aimed at evaluating the effect of a tailored exercise intervention programme in the Arterial Stiffness (AS) of old adults.
Methods: Non-randomized intervention study, enrolling 32 community dwelling old adults, aged over 65 years (mean age: 83.28 ± 8.29 years), with no prior history of major cardiovascular events. Participants were divided into two groups (intervention group—Group 2, and control group—Group 1) according to their willingness to participate in the physical training program. The exercise program included aerobic and strength components, prescribed in a personalized approach, and implemented under direct control of two experienced professionals. Aortic Pulse Wave Velocity (PWV), a measure of AS, was evaluated at baseline and 3-months after the intervention period. Clinical evaluation, anthropometric measures and blood samples were also collected in both time-points.
Results: The groups had similar clinical and demographic characteristics at baseline. After the intervention program, a significant reduction in PWV was observed only in Group 2 [F(1, 15) = 29.38; p < 0.001; η2 = 0.67]. A readjustment of the PWV age-dependent trajectory was observed, with a reduction of the absolute PWV in all age strata. A significant reduction in systolic brachial blood pressure was also depicted in Group 2 [mean difference: 12.57 mmHg; F(1, 15) = 9.46; p = 0.01; η2 = 0.39]. Parameters related to functional status, such as the handgrip strength, the self-efficacy for exercise and the activity and participation profile, were also significantly improved in Group 2.
Conclusion: Physical exercise was shown to be an effective non-pharmacological tool to positively modulate age-related decline in AS in older adults.
- Copyright
- © 2019 Association for Research into Arterial Structure and Physiology. Publishing services by Atlantis Press International B.V.
- Open Access
- This is an open access article distributed under the CC BY-NC 4.0 license (http://creativecommons.org/licenses/by-nc/4.0/).
1. INTRODUCTION
Demographic aging is a major societal challenge, imposing the need to implement strategies to promote a healthy aging and meet the challenges of this phenomenon and the additional burdens expected in the health systems [1]. In Portugal, the percentage of the elderly population aged over 65 years increased from 16% in 2001 to 21.5% in 2017, with 13.4% of the 21.5% of the older population being above 85 years of age [2]. This demographic trend brings with it several challenges, which are transversal to the various sectors of society, both in the public administration and in relation to the social inclusion of the elderly, in the expectation of an active life, in their quality and the need to guarantee individual dignity, and in the sustainability of the national health system [2–7]. On the other hand, there is still the challenge at the individual dimension, insofar as the contribution of several factors in the modulation of individual biological aging trajectories, such as social interaction, nutrition, cognitive stimulation and physical activity, is recognized [3,4].
The optimization of the intervention strategies and actions among the elderly population aiming to promote an active, inclusive and participative aging has become a priority concern, although it faces several difficulties, both in terms of the adequacy of the intervention programs to the individual needs, and in the motivation of the elderly for their active involvement in programs that imply the adoption of lifestyles often not included in their daily routines [8].
One of the most noteworthy expression of biological aging is arterial aging, due to its close association with Cardiovascular Diseases (CVD), which are responsible for most of the health problems identified in the elderly, being the leading cause of death and disability worldwide [9]. Cardiovascular risk assessment has been focused mainly on standard variables such as age, sex, concomitant diseases, Blood Pressure (BP), cholesterol, smoking habits, among others. However, other emerging risk factors have been extensively studied, with particular emphasis on the study of Arterial Stiffness (AS), established as an independent risk factor for CVD, above and beyond conventional risk factors [10,11]. In fact, arterial aging and stiffnening of the central arteries impairs the aortic buffering function, compromising the normal ventricle-arterial coupling and reducing the hemodynamic effectiveness of the heart. This is accompanied by an increase in aortic Pulse Wave Velocity (PWV) and an earlier return of the reflected waves, leading to an increase in Systolic BP (SBP) and a reduction in Diastolic BP (DBP), with a concomitant increase in Pulse Pressure (PP). Thus, the predominant phenotype of aging-related Hypertension (HT) is the Isolated Systolic Hypertension (ISH) [12]. Being a complex process, AS is influenced by several factors, such as age, BP, metabolic profile, genetics, medication, body composition, lifestyle, among others [10,11]. On the other hand, it is also known that the process of AS is accelerated by HT [13]. Frailty in the elderly has also been associated with CVD, and there is evidence that CVD predict the occurrence of frailty [14], particularly when measured by handgrip strength, a reliable indicator of frailty [15] and a marker of functional decline [16]. The measurement of PWV is a noninvasive diagnostic technique that allows an effective assessment of AS in a simple, reliable and reproducible method, being a well-established technique recommended by current guidelines [17]. The increase in AS as indexed to a higher PWV may be caused by a complex interaction of multiple factors, including the occurrence of inflammatory and atherosclerotic processes, and the presence of endothelial dysfunction [18]. Several studies have demonstrated a significant and independent association of PWV with an increased risk of cardiovascular events in different clinical settings, and particularly in the older population [19].
The identification of strategies to optimize the arterial ageing trajectory is of the utmost importance. These may include aspects such as a proper management of major risk factors, such as HT, dyslipidemia and diabetes, but also behavioral and environmental aspects, such as nutrition profile, exercise, smoking cessation, stress management, social support, pollution, and body composition management [7]. These lifestyle modifications should contribute to the reduction of BP, decrease in the pro-coagulation and inflammatory state, improvement in coronary blood flow, increase in cardiac function, and improvement in endothelial function and arterial function [18].
Therefore, physical activity should contribute to a positive modulation of the AS trajectories during ageing, particularly if administered in a personalized approach, considering the individual needs and expectations. In the present study, we address the effect of a tailored exercise programme, included in the AGA@4life model [20], with the purpose of evaluating whether this intervention can positively modulate individual arterial aging trajectories in old adults.
2. MATERIALS AND METHODS
2.1. Study Design, Population and Ethics
Community dwelling old adults were recruited from a day care center in Portugal (Association for the Defense of the Elderly and Children, Vilarinho, Portugal) and enrolled in a non-randomized intervention study aimed at evaluating the effect of a multidisciplinary intervention program on AS in old adults. Thirty-two participants with age above 65 years, with no prior history of cardiovascular and neurological disease, and physically autonomous, agreed to participate voluntarily in the study. The cohort was divided in two groups based on the degree of adherence to the physical exercise component of the intervention program: 16 participants who did not comply with the physical exercise program but fulfilled the remaining dimensions of the intervention program (cognitive stimulation, control of comorbilities, drug therapy and overall well-being promotion) were included on Group 1; 16 individuals who fulfilled the entire intervention plan, including the personalized physical exercise program were included in Group 2. The group stratification allowed to study the differentiated effect of physical activity in this population in relation to the other intervention modalities recommended in the AGA@4life project.
The study was conducted in compliance with the recommendations of the Declaration of Helsinki and approved by the Ethics Committee of the Polytechnic of Coimbra. The anonymity and confidentiality of the data collected were guaranteed. The study has strictly scientific objectives, and there were no conflicts of interest to declare. All participants signed informed consent.
2.2. General Procedure
Participants were recruited in January 2018, and a baseline individual multidisciplinary diagnostic evaluation was performed between February and March 2018. This baseline evaluation included the collection of sociodemographic information, clinical history with identification of comorbilities and ongoing therapy, determination of the physical activity profile, estimation of global cardiovascular risk and history of falls. The handgrip force was measured with a dynamometer. The self-efficacy questionnaire for physical exercise (Portuguese version) was used to assess the degree of individual confidence to perform activities [21]. The nutritional profile was evaluated by the Portuguese version of the mini nutritional assessment questionnaire [22]. The Cambridge Neuropsychological Test Automated Battery (Cambridge Cognition; Cambridge, UK) [23,24] was used to evaluate the cognitive function. Bioimpedance with an OMRON BF511 device (OMRON HEALTHCARE Co., Ltd., Japan) was used to evaluate body composition, mainly weight (kg), fat mass (%), lean mass (%) and visceral fat. Height was measured with a calibrated stadiometer. The body mass index was calculated as weight (kg)/height2 (m). The waist and hip perimeter were measured with appropriate tape in accordance to the STEPS procedure of the World Health Organization (WHO) [25]. The waist–hip ratio was calculated. The Mobil-O-Graph device (IEM, Stolberg, Germany) with a cuff of adequate diameter positioned at the level of the brachial artery was used to measure brachial BP, with the participant in sitting position, with the arm supported at the level of the heart, free of any constrictive effect of clothing and after a 5-min resting period, with participants free of tobacco and caffeine consumption for a minimum period of 2 h. Three consecutive measurements (2 min apart) were made and the average BP was used for analysis. Blood samples were collected and fasting glycaemia, cholesterol and other relevant biochemical parameters were obtained with appropriate and properly calibrated laboratory equipment. Individual cardiovascular risk was measured by the SCORE tables [26].
After the individual baseline diagnostic evaluation, a personalized intervention plan was designed and implemented for a period of 3 months. After the 3 months of intervention, participants were reassessed in all functional and physiological dimensions considered.
2.3. Intervention
The intervention program focused all the participants, being designed to correspond to the individual needs identified in the initial evaluation. This included an adapted physical exercise plan and physical therapy sessions, administered by a team of physiotherapists and physical education teachers; nutritional and therapeutic counseling by nutrition and pharmacy professionals; auditory rehabilitation activities and occupational therapy with cognitive stimulation involving professionals in the field of audiology, psychology and advanced training in the area of Education for health. Participants were divided into two groups, according to their adherence to the physical exercise plan: Group 1 with no adherence (<20% of the sessions); Group 2 with good adherence (above 90% of the sessions).
The physical exercise program was administered in a personalized way, consisting of sessions of aerobic exercise outside and laboratory sessions with a dedicated technological platform [27], with the goal of increasing aerobic capacity and strength, coordination and overall functionality. The aerobic exercise sessions followed the recommendations of the World Health Organization [28], consisting of two sessions per week with the following structure: walk at least 10 min on a flat or slightly sloping ground, joint mobilization exercises with respiratory coordination and balancing and coordination exercises (total duration per session: 1 h). The laboratory sessions were held three times a week (1-h session), including strength and balance/coordination exercises based on the Otago program [29] embedded in a technological platform using inertial and pressure sensors, feedback and exergames. The sessions were structured in an alternating pattern from Monday to Friday: laboratory session; aerobic session; laboratory session; aerobic session; laboratory session.
The intervention was implemented for 3 months after the baseline assessment, and was individually adjusted according to individual needs. Participants were further encouraged to maintain these behaviors beyond the study duration in order to ensure maximum benefit and maintenance over time.
2.4. Endpoints
Carotid-femoral PWV, a measure of aortic stiffness, was assessed with the Complior® Analyse device (Alam Medical, Saint-Quentin-Fallavier, France) according to a previously described technique [30]. The measurements were made with the participant in supine position with the neck in a slight hyperextension, and slightly rotated to the left, after a resting period of 10 min. Brachial BP was measured and entered on the Complior® Analyse software, and then signal acquisition was launched. Pulse waveforms were checked for adequate quality, and simultaneous carotid and femoral pressure curves were recorded for 15 s. The distance travelled by the pulse waveforms was measured between the two recording sites directly on the body surface, and was automatically corrected according to the equation “0.8 × direct distance”, subtracting the manubrium-to-carotid distance as previously recommended [10,30]. The aortic transit time was calculated according to the intersecting tangent algorithm, as previously recommended [10,30]. PWV was then calculated using measurements of transit time and corrected distance travelled by the pulse wave, between the two recording sites, as PWV (m/s) = distance (m)/time (s).
All measurements were performed by a highly experienced operator, with high reproducibility scores, as previously published [31], and a noteworthy concordance between invasive arterial parameters and the Complior-based pulse wave analysis method, which also been previously documented [32].
The AS was classified as abnormal according to the available reference values [10], as a value of PWV 2 standard deviations above the reference value of PWV adjusted for age and gender.
2.5. Statistical Analysis
A database was created in Excel 2016 (Microsoft Office, Redmond, WA, USA), and then imported to SPSS Statistics version 24 (IBM, Armonk, NY, USA) for statistical analysis.
The distribution of all continuous variables was checked for normality with the Shapiro–Wilks test and for homogeneity of variance with the Levene’s test. Variables with a non-normal distribution were log-transformed. Continuous variables were presented as mean ± standard deviation, and categorical variables were described as absolute and relative frequencies.
The Student’s t-test was used to compare the continuous variables obtained in the first evaluation between groups. Categorical variables were testes with the χ2 test or the Fisher exact test were used when appropriate.
A two-factor mixed-design ANOVA was used to evaluate the changes in the variables between the first and second evaluation in each and between groups. Whenever sphericity violation was identified, the Greenhouse–Geisser correction was adopted. The Bonferroni adjustment was adopted for multiple comparisons designed to locate the significant effects of a factor. Comparisons between groups in the second assessment were based on an additional ANCOVA, adjusting the comparison to the baseline values (co-variable in the model). To test if the changes in PWV were independent of changes in BP, an additional ANCOVA was performed with the PWV as the dependent variable, group as the fixed factor and the systolic BP change as the covariate. A two-tailed p < 0.05 was considered significant. The magnitude of the effects was also checked with the η2 value.
3. RESULTS
3.1. Baseline Characteristics of the Participants
The study had the participation of 32 old adults (24 women and eight men), with ages ranging from 65 to 94 years old. The main characteristics of the study population are shown in Table 1. It should be noted that no statistically significant differences were identified between groups.
| Variables | Total (n = 32) | Group 1 (n = 16) | Group 2 (n = 16) | p |
|---|---|---|---|---|
| Age, years | 83.28 ± 8.29 | 83.88 ± 7.83 | 82.69 ± 8.94 | 0.69 |
| Sex, n (%) | ||||
| Male | 25.00 (8) | 18.80 (3) | 31.30 (5) | 0.69 |
| Female | 75.00 (24) | 81.30 (13) | 68.80 (11) | |
| Weight (kg) | 67.53 ± 11.64 | 68.38 ± 10.35 | 66.69 ± 13.09 | 0.69 |
| Height (m) | 1.58 ± 0.09 | 1.58 ± 0.10 | 1.58 ± 0.08 | 0.95 |
| BMI (kg/m2) | 27.17 ± 4.49 | 27.65 ± 4.54 | 26.70 ± 4.53 | 0.56 |
| SBP (mmHg) | 156.19 ± 29.79 | 157.06 ± 30.16 | 155.31 ± 30.38 | 0.87 |
| DBP (mmHg) | 82.75 ± 19.99 | 80.63 ± 24.10 | 84.88 ± 15.36 | 0.56 |
| HR (bpm) | 72.19 ± 11.68 | 70.69 ± 11.34 | 73.69 ± 12.19 | 0.48 |
| Abdominal perimeter (cm) | 101.62 ± 8.97 | 103.73 ± 8.20 | 99.91 ± 9.45 | 0.26 |
| Waist/hip ratio | 0.93 ± 0.09 | 0.94 ± 0.08 | 0.93 ± 0.10 | 0.78 |
| Perimeter of the dominant arm (cm) | 28.33 ± 3.30 | 29.08 ± 3.16 | 27.73 ± 3.38 | 0.28 |
| Leg circumference (cm) | 34.28 ± 3.47 | 35.04 ± 3.78 | 33.61 ± 3.14 | 0.27 |
| Lean mass (%) | 21.27 ± 5.26 | 21.24 ± 5.03 | 21.31 ± 5.68 | 0.97 |
| Fat mass (%) | 26.11 ± 8.48 | 28.85 ± 7.16 | 23.38 ± 9.08 | 0.10 |
| Relation to fat mass/lean mass | 1.28 ± 0.47 | 1.42 ± 0.45 | 1.14 ± 0.47 | 0.13 |
| MNA (SCORE) | 21.82 ± 3.44 | 22.39 ± 3.50 | 21.31 ± 3.42 | 0.40 |
| Energy (Kcal) | 1281.73 ± 291.08 | 1300.79 ± 266.05 | 1265.06 ± 319.10 | 0.74 |
| Total cholesterol (mg/dl) | 183.14 ± 41.10 | 183.08 ± 51.14 | 183.18 ± 32.18 | 0.99 |
| HDL cholesterol (mg/dl) | 41.84 ± 8.24 | 41.55 ± 9.20 | 42.08 ± 7.67 | 0.87 |
| LDL cholesterol (mg/dl) | 130.70 ± 37.31 | 130.72 ± 45.92 | 130.68 ± 29.88 | 0.99 |
| Glucose (mg/dl) | 116.26 ± 63.30 | 109.33 ± 33.87 | 122.19 ± 81.57 | 0.62 |
| Creatinine (mg/dl) | 0.80 ± 0.21 | 0.80 ± 0.24 | 0.79 ± 0.17 | 0.89 |
| Uric acid (mg/dl) | 5.89 ± 1.73 | 6.04 ± 1.84 | 5.75 ± 1.69 | 0.68 |
| Urea (mg/dl) | 53.02 ± 21.96 | 54.07 ± 29.01 | 52.11 ± 14.61 | 0.83 |
| Triglycerides (mg/dl) | 148.26 ± 51.52 | 130.08 ± 38.59 | 163.84 ± 57.26 | 0.10 |
| CK (mg/dl) | 96.95 ± 62.48 | 100.85 ± 81.41 | 93.89 ± 45.68 | 0.79 |
| C-reactive protein (mg/dl) | 0.43 ± 0.65 | 0.63 ± 0.92 | 0.26 ± 0.19 | 0.15 |
| HbA1C (%) | 5.32 ± 1.87 | 5.35 ± 1.70 | 5.29 ± 2.07 | 0.93 |
| Handgrip strength (kg/F) | 14.53 ± 6.63 | 14.64 ± 3.30 | 14.44 ± 8.69 | 0.93 |
| Activities and participation profile (SCORE) | 2.01 ± 1.14 | 1.77 ± 1.12 | 2.22 ± 1.16 | 0.29 |
| Self-efficacy for exercise (SCORE) | 11.03 ± 4.30 | 11.71 ± 3.67 | 10.44 ± 4.82 | 0.43 |
BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; HR, heart rate; MNA, mini nutritional assessment; HDL, high density lipoproteins; LDL, low density lipoproteins; CK, creatine kinase protein; HbA1C, glycated hemoglobin.
Characterization of the study population
Most of the participants live alone (65.6%), and have a low level of education. Only 3.1% of the participants had smoking habits, and 18.8% had a family history of CVD. About 84.4% had HT, and the ISH phenotype was the most prevalent. Of the hypertensive patients, only 65.6% were medicated for HT, and the BP control rate was <20%. Similarly, 62.5% reported having dyslipidemia, and only 50% were on medication. Type II diabetes was identified in 25% of the cases and all were medicated with oral antidiabetic agents. No significant between-group differences were observed in all the sociodemographic and clinical variables.
3.2. Effect of the Intervention in Blood Pressure and Arterial Stiffness
Baseline PWV and brachial BP were similar in both groups. After the intervention, there was a significant reduction with a moderate effect size in PWV in Group 2 [F(1, 15) = 29.38; p < 0.001; η2 = 0.67], with no significant variations occurring in Group 1 (see Figure 1). The mean reduction in PWV was significantly higher in Group 2, even after adjusting for SBP changes, with a mean reduction of −1.06 ± 0.61 m/s versus −0.15 ± 1.06 m/s in Group 1 [Fadjusted for SBP change(1, 29) = 6.65; p = 0.001; η2 = 0.33].
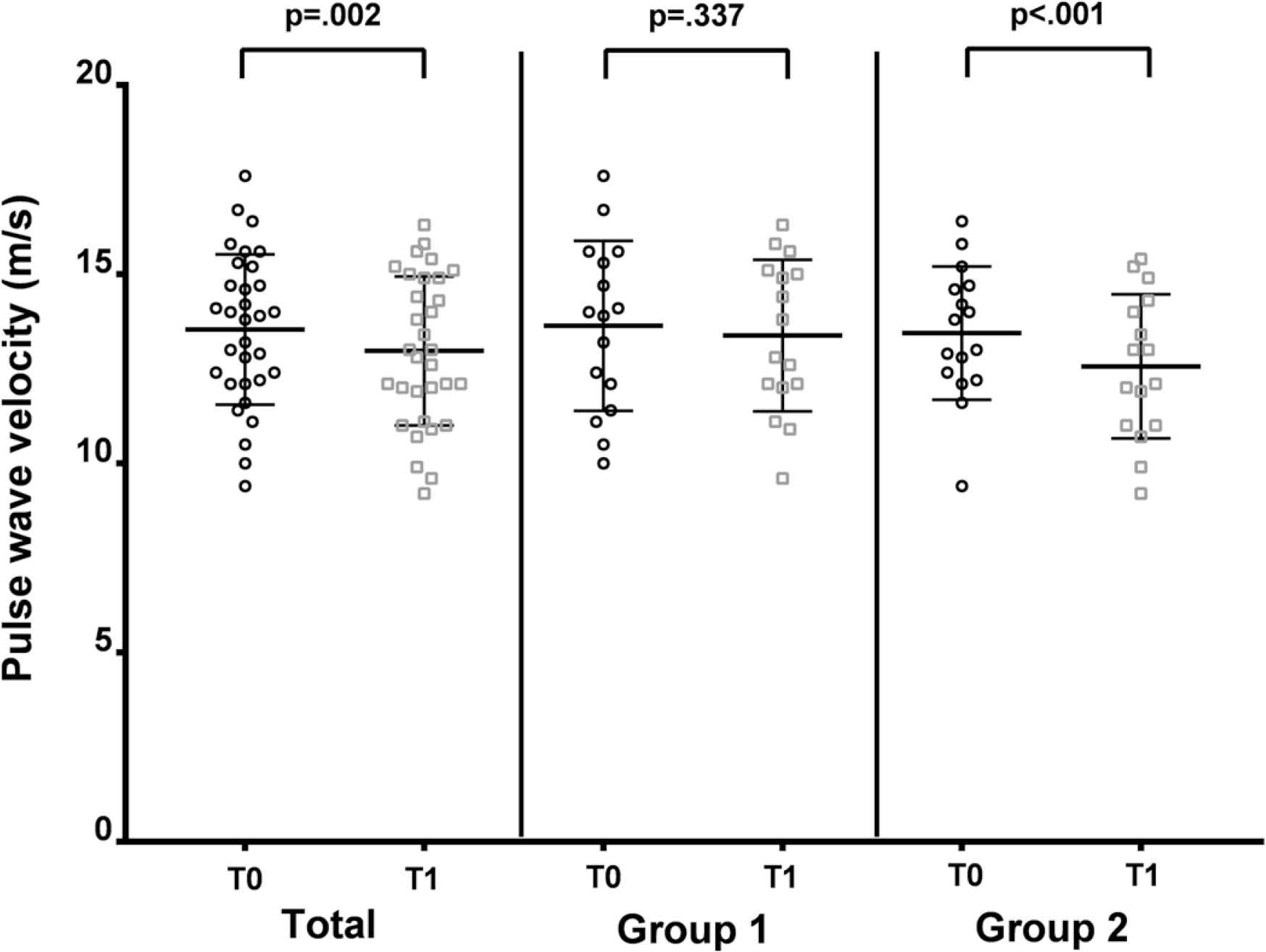
Pulse wave velocity showing a reduction from baseline (T0) to post-intervention (T1) in Group 2.
Looking at the variation of PWV with age, a readjustment of the age-dependent trajectory of PWV was identified, expressing a reduction in its absolute value throughout the age range considered in the analysis as a consequence of the intervention program (Figure 2). It is also possible to identify that the magnitude of the effect is greater the earlier the intervention (in terms of age), with the greatest reduction occurring between 65 and 80 years in Group 2, with no significant variation in Group 1.
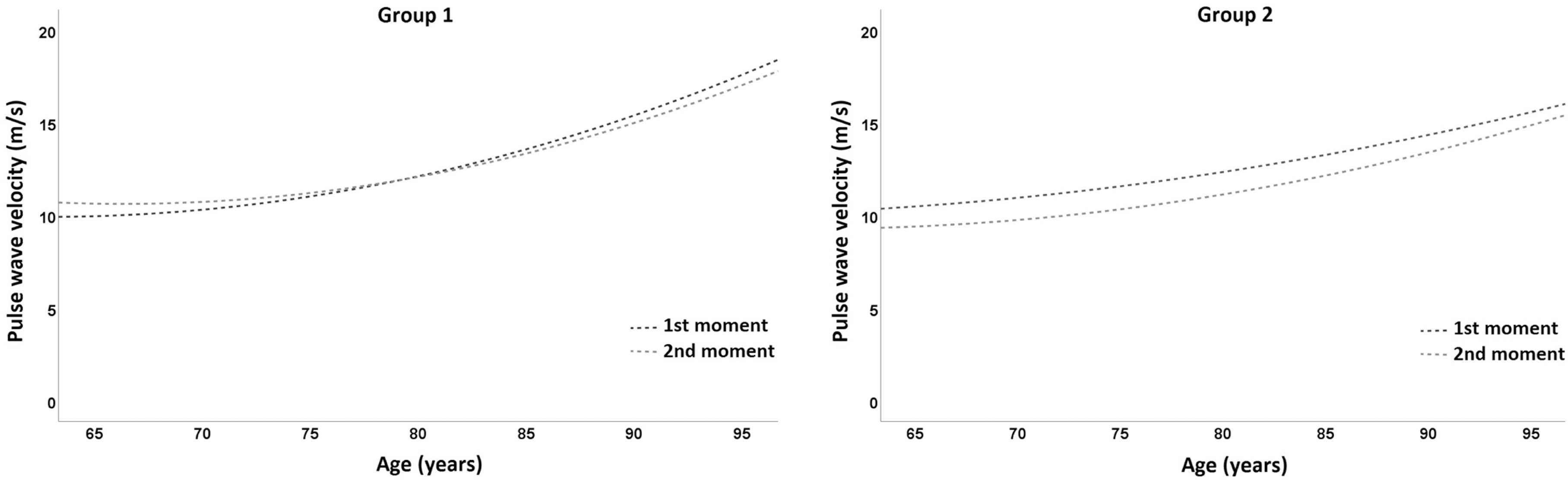
Pulse wave velocity variation with age in Group 1 (left panel) and Group 2 (right panel) in the two assessments (1st moment—baseline; 2nd moment—post-intervention).
The variation of brachial BP is represented in Figure 3. A significant reduction in SBP after intervention was observed only in Group 2 [F(1, 15) = 21.66; p < 0.001; η2 = 0.59], with a trend for a reduction in DBP also depicted in Group 2 (p = 0.076). No significant changes were observed regarding Group 1, neither for SBP nor DBP. Brachial PP also showed a significant reduction after intervention in Group 2 (p = 0.014) but not in Group 1 (p = 0.182).
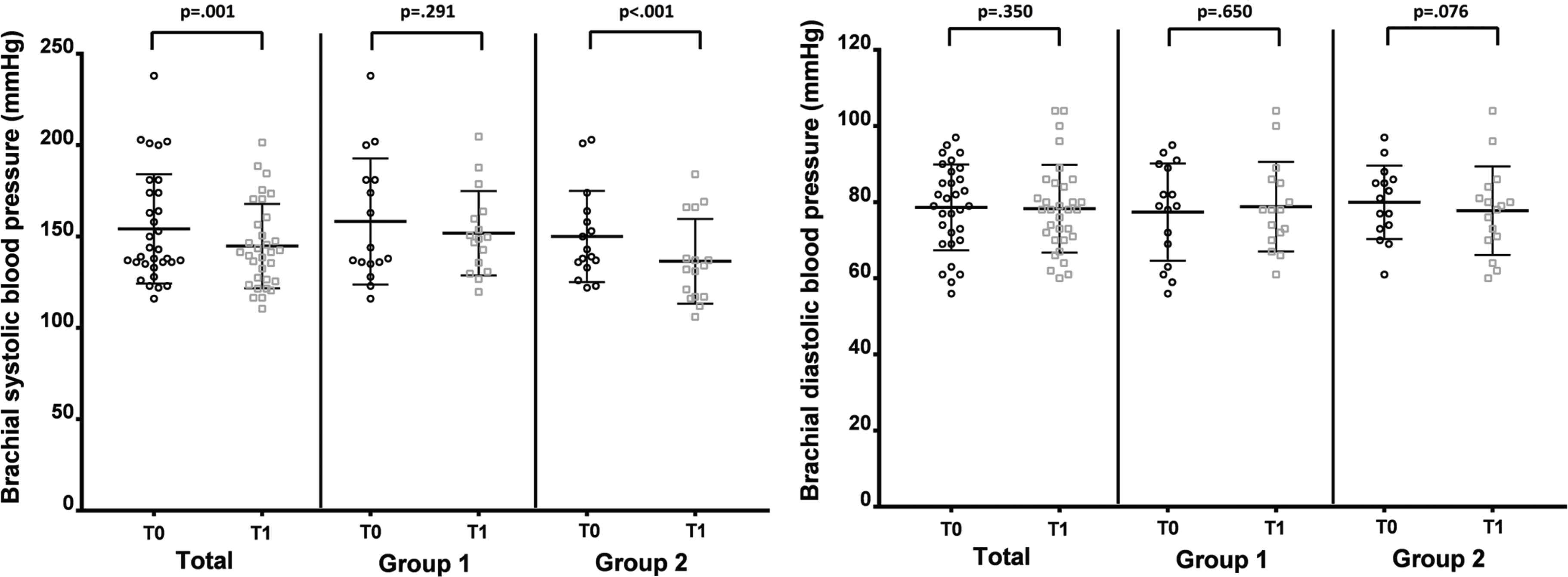
Changes in brachial blood pressure before (T0) and after (T1) the intervention in both groups. Left panel represents brachial systolic blood pressure and right panel represents diastolic blood pressure.
Regarding the nutritional profile, evaluated through the MNA, an improvement was observed in both groups as translated by a significant increase in the MNA score from baseline to post-intervention in Group 1 (p = 0.04) and Group 2 (p = 0.02). In terms of functional parameters (Table 2), significant improvements were observed only in Group 2 following the intervention programme, translating into a greater handgrip strength (p < 0.001 for Group 2; p = ns for Group 1), a better self-efficacy for exercise score (p = 0.01 for Group 2; p = ns for Group 1) and a better activities and participation profile score (p < 0.001 for Group 2; p = ns for Group 1).
| Variables | Time 1 | Time 2 | Difference (2 − 1) | p | |
|---|---|---|---|---|---|
| MNA (SCORE) | Group 1 | 22.39 ± 2.70 | 24.25 ± 1.93 | 1.56 ± 1.97 | 0.04 |
| Group 2 | 21.23 ± 3.52 | 23.17 ± 3.33 | 1.93 ± 2.77 | 0.02 | |
| Total | 21.74 ± 3.57 | 23.54 ± 2.92 | 1.80 ± 2.48 | 0.00 | |
| Handgrip strength (kg/F) | Group 1 | 12.81 ± 5.87 | 12.88 ± 6.49 | 0.06 ± 2.17 | 0.91 |
| Group 2 | 13.19 ± 5.19 | 18.38 ± 8.40 | 5.19 ± 5.49 | 0.00 | |
| Total | 13.00 ± 5.45 | 15.63 ± 7.89 | 2.63 ± 4.86 | 0.01 | |
| Self-efficacy for exercise (SCORE) | Group 1 | 10.25 ± 5.26 | 10.13 ± 5.94 | −0.13 ± 4.90 | 0.92 |
| Group 2 | 10.44 ± 4.82 | 13.94 ± 4.39 | 3.50 ± 5.76 | 0.03 | |
| Total | 10.34 ± 4.96 | 12.03 ± 5.49 | 1.69 ± 5.57 | 0.10 | |
| Activities and participation profile (SCORE) | Group 1 | 1.55 ± 1.20 | 1.51 ± 1.11 | −0.03 ± 0.75 | 0.87 |
| Group 2 | 2.22 ± 1.16 | 1.09 ± 0.52 | −1.12 ± 0.91 | 0.00 | |
| Total | 1.88 ± 1.21 | 1.30 ± 0.88 | −0.58 ± 0.99 | 0.00 |
MNA, mini nutritional assessment.
Changes in nutritional and functional variables
4. DISCUSSION
The present study evaluated the effects of a multidisciplinary intervention program on AS in community dwelling old adults, with a focus on the particular role of a tailored physical exercise program on top of other ongoing actions such as nutritional counseling, cognitive stimulation, and control of comorbidities and promotion of overall wellbeing. The health benefits of exercise have been extensively documented in all age groups, mainly due to its direct benefits on muscular strength, motor skills, cardiorespiratory fitness and overall health [1]. Accordingly, our results demonstrated significant benefits of the physical exercise intervention program, mainly a significant decrease in brachial BP, particularly in the pulsatile components (SBP and PP), and an improvement in PWV, illustrating a significant BP-independent de-stiffening effect. According to previous published studies, the practice of regular physical exercise improves cardiovascular health, by decreasing the risk of CVD and promoting lower levels of SBP [33]. Our study supports these results, considering the combined BP lowering effect with the positive arterial stiffness modulation in the group submitted to the intensive physical exercise program. The reduction of SBP may result from the combined effect of a reduction in peripheral vascular resistance, which may be due to neuro-humoral and structural responses with reduction in the sympathetic nerve activity and increase in arterial diameter due to an improved endothelial function [33], with a better arterial compliance, from which a better buffering of the left ventricle ejection wave, lesser reflected waves and better heart-arterial coupling is expected [34]. In fact, in our study the PWV also had statistically significant improvement with physical exercise, lowering from 13.44 m/s at baseline to 12.56 m/s after the intervention program. Considering that a change of 1 m/s in PWV has been associated with an approximately 14% change in the risk of cardiovascular events [11], the mean reduction of PWV by approximately 0.88 m/s after the physical exercise program translates into a significant overall cardiovascular risk reduction, further covered by the improvements also observed in BP.
One additional benefit of the physical activity program was the increased motivation for adopting healthy lifestyles as the person becomes physically more fit, which translated into a better nutritional profile as depicted in the MNA scores from baseline to post-intervention. Of course, these behavioral changes, if maintained, can contribute to delaying the onset of chronic diseases, supporting functional mobility and independent living, which is pivotal to promoting health, autonomy and social participation [1–8]. The functional gains are also of the utmost importance, and our results shown that tailored exercise training program produces a better body composition (positive modulation of the lean-to-fat body ratio), more strength (handgrip) and better self-efficacy for exercise. Recent research has identify a significant association of sarcopenia with vascular dysfunction [35], arterial stiffness and cognitive impairment [36], pinpointing a mechanistic relation between muscle mass and incident cardiovascular disease. This was also observed in the Health ABC study, an observational cohort study, in which AS, indexed to PWV, was associated skeletal muscle decline [37]. The pathophysiological link between AS and muscle mass and handgrip strength, as proxies of sarcopenia, are yet to be fully understood. Some studies suggest a link with insulin resistance [38], the accumulation of advanced glycation end products [39] and atherosclerosis progression, whilst other suggest that loss in muscle mass is associated with impaired blood vessel dynamics [37].
One added feature of the intervention program tested in our study was the inclusion of an outdoor aerobic component, following previous evidence showing additional benefits of outdoor physical activity, promoting higher levels of vitamin D, mental well-being and high emotional scores due to sensory involvement with nature [1].
There are several potential limitations to this study that must be considered. First, the non-randomized design and the small size of the studied population are important limitations that impact the statistical power of the study. Notwithstanding, the thorough characterization of each individual allowed for a comprehensive analysis of all major aspects that could introduce bias in the study, thus rendering reliable measures to the analysis. Moreover, the study was performed in real-world and daily life context providing ecological validity to the results.
In conclusion, the major findings of this study supports a tailored physical exercise intervention as an effective non-pharmacological tool to positively modulate age-related decline in arterial function in old adults, particularly when prescribed in a personalized approach and with a multicomponent structure, as foreseen in the innovative AGA@4life model, lowering BP and PWV, providing enhanced motor control, better functionality, balance and coordination, and improving overall health and well-being.
CONFLICTS OF INTEREST
The authors declare they have no conflicts of interest.
AUTHOR CONTRIBUTIONS
TP were responsible for the study conceptualization. Data curation and formal analysis were carried out by TP, FS and IC data curation, formal analysis and writing (original draft) the manuscript. Funding acquisition, project administration and supervision of the project were carried out by TP. Review and editing of the final manuscript were done by TP, FS and IC.
FUNDING
This work is co-financed by the
Footnotes
REFERENCES
Cite this article
TY - JOUR AU - Telmo Pereira AU - Fabiana Santos AU - Inês Cipriano PY - 2019 DA - 2019/12/04 TI - Effects of a Personalized Physical Exercise Program in the Arterial Stiffness in Older Adults JO - Artery Research SP - 57 EP - 64 VL - 25 IS - 1-2 SN - 1876-4401 UR - https://doi.org/10.2991/artres.k.191126.001 DO - 10.2991/artres.k.191126.001 ID - Pereira2019 ER -
