Prevalence, Risk Factors, and Epidemiology of Food-borne Botulism in Iran
- DOI
- 10.2991/jegh.k.200517.001How to use a DOI?
- Keywords
- Botulism; Clostridium botulinum; epidemiologic surveillance; laboratorial diagnosis
- Abstract
Background: Botulism is a severe neuroparalytic disease caused by toxins produced by several Clostridium species. This work presents the surveillance results of botulism in Iran, with the distribution of the cases by regions and by vehicle of transmission.
Methods: We describe the findings of the Centers for Disease Control and Prevention (CDC) surveillance on 2037 suspected cases of food-borne botulism during 2007–2017.
Results: A total of 252 (12.3%) cases were confirmed to food-borne botulism. The mean annual incidence per 100,000 Iranian Natives was 7.1 cases for male individuals and 3.3 cases for female individuals. All botulism events were confirmed to be foodborne. The most commonly implicated food was home-prepared traditional processed fish product, followed by the consumption of commercially canned products and non-pasteurized dairy products. Forty-eight (19%) fatal botulism were reported which, the case-fatality rate declined from 4.5% to 0.7% during the study period.
Conclusion: Laboratory-based diagnosis of botulism is an imperative procedure to elucidate cases, particularly food-borne botulism, to identify toxins in food and confirm clinical diagnosis, helping sanitary control measures. In addition, educational materials related to botulism prevention should be disseminated to different communities.
- Copyright
- © 2020 Atlantis Press International B.V.
- Open Access
- This is an open access article distributed under the CC BY-NC 4.0 license (http://creativecommons.org/licenses/by-nc/4.0/).
1. INTRODUCTION
Botulism, a potentially fatal neuroparalytic disease, is caused by highly potent neurotoxins produced by the Gram-positive, anaerobic spore-forming bacteria, Clostridium botulinum, Clostridium barati, Clostridium butyricum, Clostridium sporogenes and Clostridium argentinense [1,2]. The clostridial species can produce up to seven antigenically distinct exotoxins (A, B, C1, D, E, F and G), which are differentiated serologically by specific antitoxin. Types A, B, E, and more rarely F, are responsible for most human cases [3]. Botulinum Neurotoxin (BoNT) blocks evoked quantal acetylcholine release from motor ending at all peripheral cholinergic synapses, producing a profound but transient muscle flaccid paralysis [4]. If remained untreated, a severe case of botulism leads to death of the patient due to paralysis of the respiratory muscles [5].
Food-borne botulism, the most widely recognized form, is caused by eating food containing BoNT [6]. Since C. botulinum is ubiquitous in the environment, spores routinely contaminate food and survive by standard cooking practices that do not surpass 100°C. Clostridium botulinum produce botulinum toxin under specific conditions that incorporate an anaerobic, low-acid and -salt environment. Home-preserved foods regularly achieve these conditions, and therefore, present a high risk for botulism.
Regardless of the form or serotypes involved, nonetheless, human botulism is a medical crisis that requires rapid intervention. Because prompt administration of antitoxin can reduce the severity of the disease [7], the choice for treatment depends on clinical diagnosis and epidemiologic data, without laboratory confirmation. Along the world, there are many reports on food-borne botulism due to the consumption of traditional salted fish, dairy products such as cheese and kashk, and canned foods in Iran [8,9]. Different sorts of fish; smoked, salted, and canned, additionally canned green beans and cucumber are causative agents of food-borne botulism recorded in some medical centers in Tehran and various regions of Iran, between April 1984 and August 1994 [10]. Examination of food-borne botulism incidents provides valuable data in regards to implicated foods and conditions resulting in toxin formation. In order to identify outbreaks as rapidly as possible, the Centers for Disease Control and Prevention (CDC) maintain an intensive National Botulism Surveillance System, provide epidemiologic consultation and laboratory diagnostic services, and administer the antitoxin release process. In order to collect and analyze data, the Iranian national care system for botulism was designed and implemented in 2003. In addition, the epidemiological survey and immediate report of this disease to the health centers are underlined by the CDC. This study aimed to retrospectively investigate the changes of epidemiology, clinical and laboratory diagnosis of all suspected and laboratory-confirmed cases of food-borne botulism in Iran over a period of 11 years (2007–2017) following major political and economic changes, addressing the question of whether botulism is still an important health concern in Iran.
2. MATERIALS AND METHODS
The information exhibited in this study refer to the cases of botulism reported to the Iran’s CDC within periods of 2007–2017. The present reporting system provides for the immediate notification of any case of botulism, even if only suspected, from the physician to the Local Health Authority; the flow proceeds toward the Region, the Ministry of Health (MOH), the National Institute of Health. For the transmission of the results of laboratory testing, either positive or negative, the similar flow is done. The diagnosis of botulism was verified on epidemiological data and a clinical evidence of severity according to Public Health Agency of Canada. These signs and symptoms were: (1) Problems of accommodation (visual impairment); (2) dryness of the mouth (dry syndrome); (3) retarded intestinal transit; (4) dysphagia with liquid and/or solid meals; (5) persistent constipation; (6) urinary symptoms; (7) asthenia; (8) respiratory paralysis; and (9) peripheral paralysis and impossibility of oral feeding. Clinical samples include serum (10 mL), vomitus or gastric secretions, stool and suspect foods in original containers that were evaluated for toxin or C. botulinum [8]. Botulinum toxin was detected by bioassay. Toxin detection and identification by toxin neutralization tests in mice (mouse bioassay) was performed in a limited number of public health labs [11]. In addition to cases with laboratory confirmation, persons with botulism who were epidemiologically linked to a laboratory-confirmed case were considered to have confirmed, and thus reportable cases.
Demographics and disease information including age, gender, occupation, place of residence (urban or rural), town/city, type of toxic agent, month, season outcome (recovery or death), antitoxin administration, signs and symptoms were documented by a baseline questionnaire [12]. Every one of this information, together with the lab and microbiological analysis results are collected and stored in a special database, which transmits all information to the MOH yearly. The gathered information was coded, computerized and analyzed utilizing SPSS software (ver 22, Chicago, Illinois, USA) by univariate examination with t-tests and Chi-squared, as appropriate. For all tests, significance was defined as p < 0.05.
3. RESULTS
3.1. Demographics and Incidence
During the period 2007–2017, 2037 cases suspected to food-borne botulism were reported, including 252 (12.3%) confirmed and 743 (36.4%) cases were remained suspect to botulism (Table 1). Almost all patients (99.3%) were identified as Iranian Native. The mean annual number of cases was 185.1 (range, 16–444 cases per year). About 44% (914 of 2037) of the reported cases and 18.6% (47 of 252) of confirmed cases were female. Most of the reported cases were laborers and people with limited formal education 672 (33%), housewife 635 (31.2%) or students 334 (16.4%). The next largest groups were: children 205 (10.1%), intellectuals and office workers 109 (5.4%). However, most of the confirmed cases were housewife 66 (26.1%), children 54 (21.4%), farmer workers 37 (14.6%) and students 32 (12.6%). The profession for other patients was not specified in the medical records. Overall median age was 28.4 years (range, 2 months to 88 years). The overall and sex-adjusted mean annual cases of foodborne botulism by period are shown in the Figure 1.
| Year | Confirmed case | Suspected case | Fatality rate |
|---|---|---|---|
| No. (%) | No. (%) | No. (%) | |
| 2007 | 0 (0) | 16 (100) | 1 (6.2) |
| 2008 | 2 (3.2) | 40 (64.5) | 2 (3.2) |
| 2009 | 1 (1.1) | 84 (98.8) | 1 (1.1) |
| 2010 | 45 (16.7) | 164 (61.1) | 10 (3.7) |
| 2011 | 83 (18.6) | 279 (62.8) | 26 (5.8) |
| 2012 | 24 (16) | 85 (56.6) | 4 (2.6) |
| 2013 | 17 (15.1) | 70 (62.5) | 4 (3.5) |
| 2014 | 27 (10.3) | 2 (0.7) | 0 (0) |
| 2015 | 18 (5.9) | 1 (0.3) | 0 (0) |
| 2016 | 24 (13.4) | 1 (0.5) | 0 (0) |
| 2017 | 11 (6.9) | 1 (0.6) | 0 (0) |
| Annual mean | 22.9 | 67.5 | 4.3 |
| Total | 252 | 743 | 48 |
Number of confirmed and suspected cases of foodborne botulism by year, Iran, 2007–2017
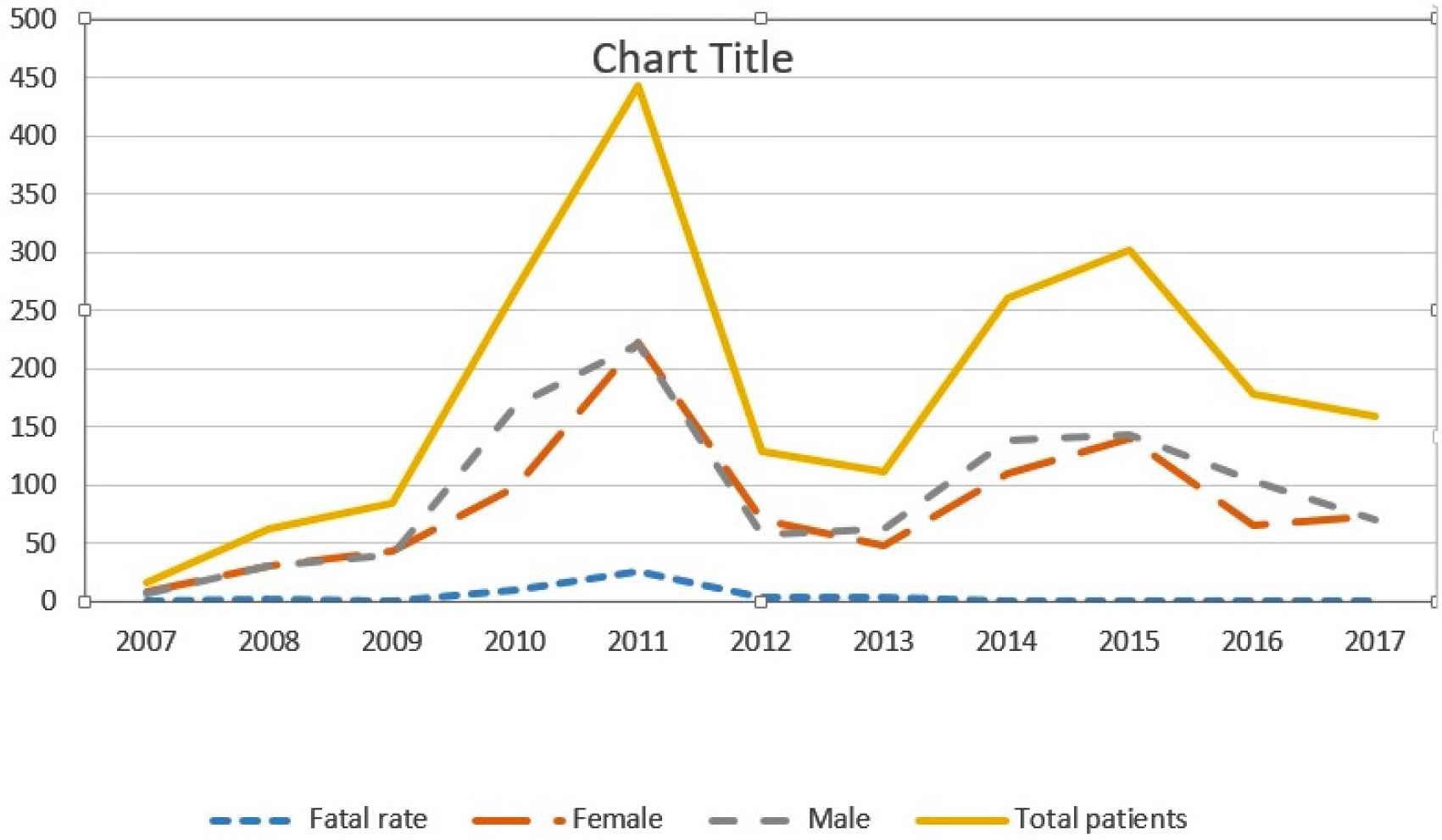
Overall and sex-adjusted mean annual cases of foodborne botulism Iran, by period.
During 2007–2017, the mean annual incidence per 100,000 Iranian Natives was 7.1 and 3.3 cases for male and female individuals, respectively. The incidence was not associated with age group (p > 0.05); patient’s age increased from 2007–2012 (median age, 25.2 years) to 2013–2017 (median age, 31.5 years; p < 0.05). About 35% (398 of 1117) suspected cases occurred in urban areas accounting for an incidence rate of 4.2 patients per 100,000 urban inhabitants. Data is not available for other 920 individuals.
3.2. Morbidity and Mortality
The percentage of confirmed cases hospitalized during 2007–2012 (88%) was similar to the percentage of patients hospitalized during 2013–2017 (87%). During the study period, 81% of the botulism patients had received Intravenous (I.V) or Intramuscular (I.M) antitoxin types A, B, and E. These administration have occurred at a median of 1 day (mean, 1.37 days) after illness onset when botulism was initially suspected and a median of 3 days (mean, 3.11 days) when botulism was initially misdiagnosed (p < 0.05). In addition, 38% (96 of 252) had received mechanical ventilatory support. Data regarding underlying medical conditions, adverse reactions to antitoxin, and overall duration of illness were unavailable.
The mouse bioassay is done in a limited number of public health labs. From the time that mice are injected, final results may not be available for 24 or 48 h. Likewise, all management decisions and initial public health interventions are determined based solely on clinical diagnosis. Botulism was suspected at the initial clinical presentation for 79% of the cases. However, initial misdiagnosis occurred in 31% of cases. The clinical pattern was mainly characterized by dry mouth 154 (61.1%), vomiting 151 (59.9%), double and/or blurred vision 92 (36.5%), constipation 75 (29.7%), drooping eyelids 74 (29.4%), dysphagia 59 (23.4%), abdominal pain 58 (23.0%), slurred speech 47 (18.6%), and mydriasis 34 (13.4%). When botulism was not suspected, the initial diagnosis was frequently a gastrointestinal illness, encephalitis respiratory illness, or signs of poisoning with chemical pesticides.
About 19% (48 of 252) fatal botulism were reported during 2007–2017, which was not significantly different among different age groups (Table 1). Within the 11-year study period, the case-fatality rate declined from 4.5% in 2007–2011 to 0.7% in 2012 2013 and then remained zero during the remaining years (Table 1). However, since mortality data were gotten from the lab databases that do not officially keep mortality records, these data should be interpreted with caution. Data regarding the association between type of botulism and case fatality rate are not available.
3.3. Implicated Foods
All botulism events include confirmed or suspected cases were confirmed to be foodborne. The most commonly implicated food was home-prepared traditional processed fish (smoked fish, salted fish, ham, bacon, blood pudding, mosaic salami, sausage), accounting for 34.1% events. Other implicated food was commercially canned fish (28.6%), fish spawn (10.5%), diary product (10.1%), vegetables and home-prepared legumes (9.7%), cottage cheese (5.9%), and canned fruits (1.1%). Botulism events varied by season; most occurred in the spring (37.4%) or summer (33.0%), compared with winter (16.4%) and autumn (13.1%). About 18% of patients had a history of travel to other cities or provinces.
4. DISCUSSION
Food-borne botulism remains a potentially fatal public health risk among Iranian Native persons [13,14]. The annual mean of 26.2 confirmed botulism cases during 2007–2011 was greater than reported cases during the 2012–2017 period; also, the case-fatality rate dramatically decreased from 15.8% to 3.1%, respectively. The recent decrease illustrates the impact of statewide prevention efforts and increased awareness of symptoms in high-risk communities, which leads to prompt medical attention and the administration of antitoxin. Across time, botulism incidence among Iranian individual remained higher among the male population, and occurrence of outbreaks peaked during spring and summer [15]. This higher rate of botulism poisoning in men is probably because of the cultural differences between them; men are more willing to eat canned foods and are less likely to heat food [16]. The seasonality of botulism outbreaks has been described previously [17] and is probably related to warmer temperatures and seasonal dietary variations.
Food consumption practices were also assessed in our survey. The primary cause of the disease was the consumption of home-prepared traditional processed fish and fish product, followed by the consumption of commercially canned products and non-pasteurized dairy products. Consumption of these foods either cooked or raw may contribute to food-borne toxicity in populations. A retrospective survey on food-borne botulism poisoning cases at north of Iran during 2001–2006 showed that conserved foods (38.7%), salted caviar (32.3%), and fish (16.3%) were the main causes of botulism, respectively [16]. Additionally, it was mentioned that anti-botulism is administered too hasty for some patients. Although, usually, in the empirical disease management, there was no escape from this snaps. Another survey of C. botulinum toxins in Iranian traditional food products indicate that some Iranian traditional foods (cheese and salted fish) may contaminated by different types of C. botulinum [8]. In their study, toxins were detected in 4.58% of analyzed samples (6.36% of salted fish samples and 3.51% of cheese samples). In a report from the Pasteur Institute, 27 people in Qazvin province were affected after consuming traditional cheese and botulinum toxin (type A) was detected in 10 patients [18]. Hydarnia and Memarbashi [19] showed the presence of botulinum toxin in traditional kashk (kind of dairy product in Iran). After this study, the Iranian Health Ministry decreed that all kashk for sale in Iran had to be pasteurized.
During 2002–2007 (6-year period), 260 cases of botulism were detected in Iran, of which 38 cases were related to the consumption of traditional dairy products and 88 cases to the consumption of salted sea food [20]. In the similar period, another examination was performed to determine the rate of food-borne botulism illness [13]. It was reported that among 341 suspected cases of botulism, the most and the least cases related to the years 2007 (28.28%) and 2003 (12.6%), respectively. Among the regions, Guilan lead the cases with 90 occasions while the least amount related to West Azerbaijan with four instances. Salin fish and spawn (31.08 %) were the most botulism producing factors. One of the most predominant causes of food poisoning (53.07%) was providing and consumption of canned foods at home. Additionally, A and E types with the rate of (18–47%) and (13.7–41.3%), respectively were the most common causes of engendering botulism food poisoning [13]. This kind of food poisoning had an escalation probably because of paying more attention to record and reports of the occurred cases in recent years in comparison with what was used to be. It appears to be important to suggest the restaurants of this regions that serve fish and salted caviar in their menu list of foods to observe sanitation rules to prevent botulism due to fish contamination and the greater role of aquaculture in causing this disease. It is also necessary to hold courses on early detection and on time referral for physicians. Some measures like general health education, not using unsanitary and traditional food processing, heating sufficient while consumption, not using unpasteurized dairy product, and regular supervising by health inspector can prevent claimed issue.
Botulism is relatively rare, thus some physicians are unfamiliar with the disease. Therefore, patients with botulism can be misdiagnosed as having other illnesses such as Guillain-Barré Syndrome, Miller-Fisher Syndrome and myasthenia gravis, resulting in delays in the administration of botulinum antitoxin and increased mortality [21]. Although botulism is typically rare, fatality can be high if the disease is not treated immediately. The high mortality rate is related to delayed clinical diagnosis, and respiratory failure is a major cause of death. A significant reduction in mortality is observed when patients are treated in hospital units with intensive therapy [22].
Laboratory-based diagnosis of botulism is a fundamental basis for elucidating cases, particularly food-borne cases, affirming the clinical diagnosis and identifying toxins in food, allowing for epidemiologic surveillance for sanitary control measures. Although micro-organism culture methods can be applied for the isolation of viable cells of C. botulinum in clinical samples or food, they are not adequate to confirm the reason of illness, requiring interpretation of the results related to case definition in the epidemiologic investigation. The demonstration of C. botulinum in patient stool samples or in cultures of wound material is generally satisfactory for the diagnosis of adult botulism syndromes and is considered definitive for the diagnosis of infant botulism [3].
Preparation of appropriate samples (depending on the type of botulism) before administration of antitoxin can lead to an increase in the number of positive cases of the specimens. It should be noted that compared with the industrialized countries, the percentage of positive samples in Iran is very low, showing that there are serious problems in the following areas: (1) Weakness in identifying suspected cases of botulism; (2) preparation of required samples before administration of antitoxin; (3) the inadequacy of the samples in terms of the quantities required for testing; (4) delay in the timely delivery of samples and failure to comply with standard conditions in their submission; (5) preparation and provision of anti-toxins A, B, and E are another common problem that we face in the treatment of patients. However, in recent years, improved national care system for botulism has increased the speed and accuracy of diagnosis and increased awareness-based care has also led to a reduction in incidence, rapid diagnosis of the onset and reduction of death.
In conclusion, the results of this investigation show that different types of C. botulinum may be present in Iranian traditional food products. Since these products are consumed raw or under-cooked, consumption of them may pose a risk of food-borne botulism in the Iranian population. Improved methods to reduce the risk for food-borne botulism and support behavior change should be explored. In addition, educational materials related to botulism prevention should be disseminated to different Iranian communities each spring, with messaging that targets the population and younger persons.
CONFLICTS OF INTEREST
The authors declare they have no conflicts of interest.
AUTHORS’ CONTRIBUTION
MRMK and BE participated in study supervision, design of the study, and critical revision of the manuscript for important intellectual content. AZB and MR carried out the data collection and drafted the manuscript. MMG and FS participated in design of the study and critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript.
ACKNOWLEDGMENTS
The authors would like to acknowledge the kind collaboration of all the staff at provincial and district health centers and lab technicians in different provinces for data collection. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Footnotes
REFERENCES
Cite this article
TY - JOUR AU - Mohammad Reza Montazer Khorasan AU - Mohammad Rahbar AU - Abed Zahedi Bialvaei AU - Mohammad Mehdi Gouya AU - Fereshte Shahcheraghi AU - Babak Eshrati PY - 2020 DA - 2020/05/25 TI - Prevalence, Risk Factors, and Epidemiology of Food-borne Botulism in Iran JO - Journal of Epidemiology and Global Health SP - 288 EP - 292 VL - 10 IS - 4 SN - 2210-6014 UR - https://doi.org/10.2991/jegh.k.200517.001 DO - 10.2991/jegh.k.200517.001 ID - Khorasan2020 ER -
