Optimality, cost minimization and the design of arterial networks
- DOI
- 10.1016/j.artres.2015.01.001How to use a DOI?
- Keywords
- Circulation; Design; Optimality; Shear stress
- Abstract
The arterial circulation acts as a network to deliver nutrients and oxygen to cells. The design of the cardiovascular system is subject to a variety of constraints and costs. It has been postulated that the design of the arterial network might be understood in terms of the need to minimize competing ‘costs’ within the context of physical or material limits to the system. These designs can also be envisaged as being subservient to space filling or fractal considerations. The signalling mechanisms underlying these designs remain to be fully characterized although shear stress, wall tensile stress and metabolic stimuli are likely candidates. I will also review evidence that deviations from a minimal cost condition or optimal design may provide both a measure of disease severity and insights into the underlying disease mechanism.
- Copyright
- © 2015 Association for Research into Arterial Structure and Physiology. Published by Elsevier B.V. All rights reserved.
- Open Access
- This is an open access article distributed under the CC BY-NC license.
Introduction
In branching cones the living web expandsa Lymphatic ducts, and convoluted glands; Aortal tubes propel the nascent blood, And lengthening veins absorb the refluent flood;
The aim of this paper is to review ideas regarding the design of arterial networks in relation to optimal design and cost minimization. I will also discuss some evidence that disease is associated with deviations from an ‘optimal’ design. I will not review the genetic, epigenetic, or signalling mechanisms putatively involved in establishing and maintaining optimal design or issues related to optimal coupling of the heart and vascular system. For these topics readers are referred to other articles.1–6
Design of arterial networks
The idea that morphology and function are causally interrelated can be traced back at least to Hellenistic philosophy7,8; however attempts to make quantitative links between morphological design and function based on mechanistic arguments or analysis emerged in the Enlightenment, following the work of Galileo, Borelli, Newton and Harvey.9
In 1515 Leornardo da Vinci described tree boughs as preserving cross-sectional area at branchesb, but as far as I know the first attempt to quantify relationships between blood vessels at bifurcations in an arterial network was made by James Keill in 1708. Keill made anatomical measurements of arteries from dog, calf and man with the aim of calculating ‘the Quantity of Blood in the Humane Body’. He found that the ratio of vessel cross-sectional areas at a bifurcation was typically 41616:52126c (i.e. 1: 1.25) and used this ratio in combination with geometric scaling laws to give crude estimates of total blood volume and blood flow velocity in capillaries. Woldenberg10 has provided a detailed critical description of Keill’s work on the arterial network, and his relationship to the English ‘iatromechanists’,d and to other scientists, such as Hales and Young. Young refers to Keill’s data in his 1808 Croonian Lecture11 where he assumes a consistent increase in area of 1:1.26 at each arterial bifurcation. Young makes no comment on the possible significance of this relationship (1:21/3), although it seems unlikely that it could have escaped his notice.12 Roux, in his doctoral thesis later in the 19th century, undertook a detailed study of the relationships between diameters and the angles subtended by arteries at bifurcations.13 He concluded that ‘the shape and direction of the lumen of the blood vessels at their branch points is mainly determined by the action of hydrodynamic forces’. Roux is probably best known the founder of the Entwick-lungsmechanike, a ‘Kantian Mechanist’ programme for embryology and development.14 Roux envisaged that development was shaped by the interaction between forces and ‘Darwinian’ selection within an organism operating at a cellular level.15 Roux’s views were very influential in the late 19th and early 20th century and contributed to a greater integration of physics and mathematics into biological analysis.16 In 1901 Richard Thoma17 proposed that the size of arteries depended on the velocity of blood flow in the vessel. He proposed that the diameters between parent and offspring branches conformed to an exponential relationship
In the first pair of papers20,21 Murray aimed to find physical laws that described the organization of the vascular system in relation to oxygen transport and exchange at the capillary level. He envisaged this as ‘a problem of maxima and minima’ and employed the idea of two competing economic factors: the cost of blood flow (i.e. powerf expended) and the cost of the blood volume. Using an assumption of Poiseuille flow and that the cost of the blood volume per unit length was proportional to the area of the vessel, Murray calculated that for maximal efficiency (in terms of blood flow and volume) blood flow should be proportional to the cube of the radius of the vessel, r, hence for a bifurcating network minimization of cost would be achieved if
Murray commented that Hess had neglected the conditions in the other branch (or continuing segment of the parent artery) on his analysis and that his conclusion was therefore incorrect.
Another comprehensive treatment based on the minimum energy principle was undertaken by Cohn23,24 in 1954. Cohn argued that four factors should be optimised: 1) the size of the aorta; 2) capillary dimensions and the volume of tissue supplied by a capillary; 3) the connecting system between the aorta and the capillaries, and 4) the total resistance of the system to flow. The size of the aorta was considered to be constrained by the need to avoid turbulence; the capillary dimensions and capillary density were determined by diffusional considerations and the size of the erythrocyte; while the connecting network was considered to be constructed to optimise space filling while minimising resistance. For simplicity, Cohn assumed that branches were symmetrical and minimized resistance on the basis of Poiseuille flow. He also assumed that the mass of the blood vessel wall was determined by Laplace’s relationship for wall tension. Cohn then derived an ‘optimal’ relationship of
Taylor25 developed Cohn’s arguments and applied them to pulsatile flow in elastic arteries in a rarely citedg but interesting paper. Taylor accepted the assumptions of minimum energy expenditure in terms of blood flow and blood volume but also argued that it would too costly in terms of cardiac work to allow the small arterial terminations to be impedance matched to the proximal conducting elastic arteries; hence reflections were inevitable. Essentially this constraint appears to be an extension of the minimum volume argument, although Taylor does not make it explicit. Taylor further assumed that optimality considerations required global impedance (the complex ratio of pressure to flow rate) to be stable over a range of frequencies and minimized. He drew two interesting and important conclusions from his analysis: 1) that if the distance to the “average reflecting site” of the scattered terminations were greater than the quarter wavelength of the fundamental harmonic then out of phase reflections from terminations would tend to cancel out and 2) if the wave speed of the network increased progressively outward along the paths connecting the heart to the terminations this would tend to ‘uncouple’ the termination and minimise the impact of reflections. Taylor concluded that the branched anatomical design of the arterial network allowed the heart to ‘see’ the proximal distensible region while keeping the overall compliance of the system low and minimizing the influence of reflections on the heart. Although not couched in these terms Taylor described a system that from the heart’s perspective looks like a ‘Windkessel’ despite the presence of reflected waves. More recently Parker et al.26 have used an analogous power minimization argument to propose that reservoir pressure, which roughly equates to Windkessel pressure, represents a minimum power condition for the circulation and that the difference between total and reservoir pressure (excess pressure) might be used as an indicator of non-optimal circulatory performance. Consistent with this suggestion excess pressure has been reported to predict cardiovascular events independent of conventional risk factors.27
Zamir28 recognised that the optimality approaches outlined above lacked any plausible mechanism by which global optimality could be implemented. In other words how could a single bifurcation sense what was going on in billions of other bifurcations or the total volume of blood in the system? Zamir29 noted that under conditions of Poiseuille flow the shear stress, τ, was given by
In 1981 Sherman12 published an influential review that summarized much of the work up to that time and examined the extent to which Murray’s law fitted experimental data. He concluded that the fit was good, although not perfect, and he made some important points: 1) Murray’s law might apply to inanimate systems if a trade-off between power and volume were made at some point during construction or development; 2) Murray’s law would only apply to systems where flow conductance was proportional to r4 (i.e. Poiseuille flow); 3) changes in diameter would have to be coordinated across a large region in response to dynamic change.
Subsequent authors addressed the impact of turbulence34 and viscosity across the network34,35. Several studies looked at the predicted costs associated with deviation from optimality.34,36–38 These studies showed relatively shallow cost functions for small deviations from optimal which became much steeper with large deviations. In general the observed deviations seen in experimental data incurred costs that were small (<10%). Taken together these studies and others (reviewed in 39) provided fairly substantial broad support for Murray’s law in a range of fluid conducting systems where Poiseuille flow was present and these data were consistent with a role for shear stress in maintaining this relationship.
West et al.40 dealt with large arteries in an important but controversial41–43 paper on allometric scaling. They proposed that the branch exponent for large elastic arteries with pulsatile flow should be 2 (area preserving) and that there should be a rather abrupt ‘step-like’ transition to the area-increasing (shear-preserving) relationship predicted by Murray in smaller arteries. It was assumed that branching followed a fractal-like space filling paradigm (see below) and that capillaries were size-invariant units. Womersley’s linearized solution to the Navier–Stokes equations was used with the assumption of a thin wall and incompressible fluid. Under these assumptions the impedance, Z is given by
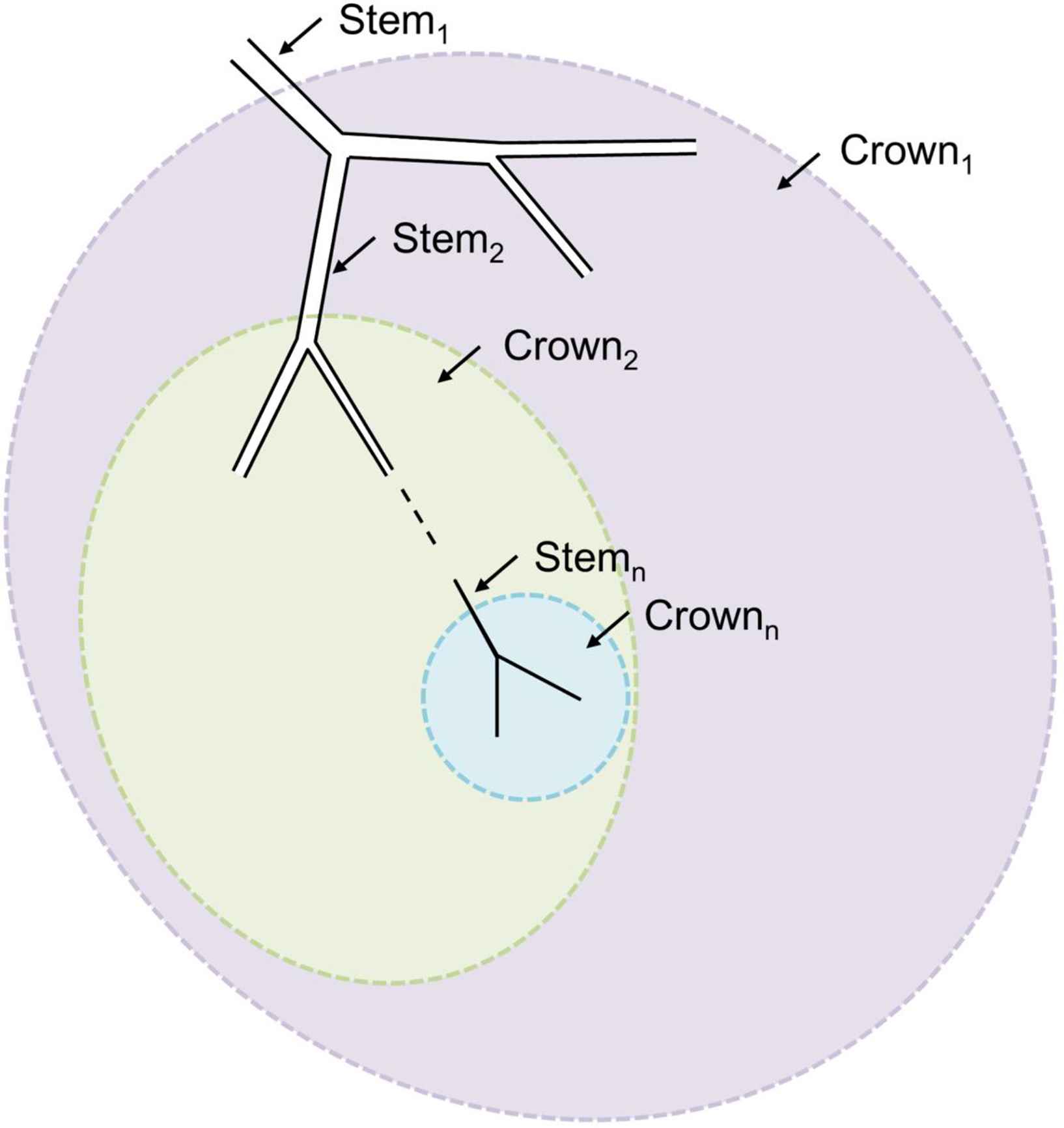
Mechanisms of optimality
Sherman12 had considered that coordination of network behaviour would be difficult to achieve following dynamic change in a network of arteries. Griffith and colleagues50,51 however provided experimental evidence of a mechanism that could account for this. They used perfused rabbit ears with five generations of branches (G0 to G4) visualised by microangiography. They showed that flow-dependent release of endothelium derived relaxing factor (EDRF; probably nitric oxide (NO) in this experimental system) maintained diameters of different generations of resistance arteries close to Murray’s predicted optimum, either under resting conditions with myogenic tone, or following vasoconstriction with serotonin.50,51 When EDRF was inhibited with haemoglobin, branching exponents deviated markedly from 3 with the average junction exponent being ∼6 in the presence of serotonin. Similar findings were made in the human retina under resting conditions in vivo when NO synthase was inhibited by NG-monomethyl-
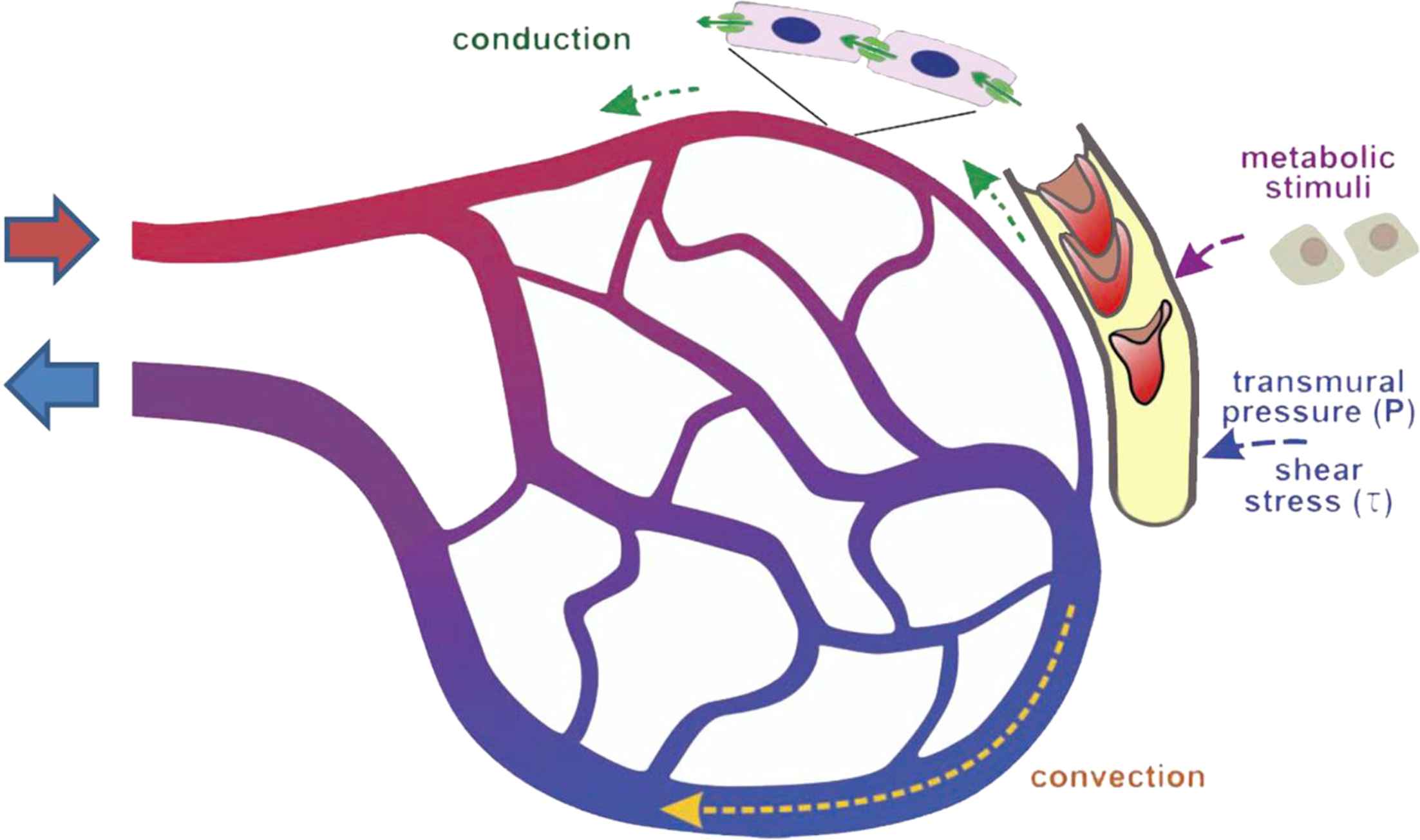
The mechanisms that might account for the closer adherence of large elastic arteries to an area-preserving branch exponent (x ∼ 2) are less well understood. However, one might speculate that limited access of NO and other endothelium-derived mediators into the arterial wall,61 wall circumferential stress62 and/or the effects of flow separation and complex flow63 could be important factors.
Fractals
Fractals were first applied to arterial design in the 1980’s. At first sight fractal analysis appears to differ from cost-effectiveness analysis, although in fact the two can be linked through scaling considerations.40,64 The term fractal was coined by Mandelbrot to describe self-similar structures with a fractional (non-unity) dimension and devoted some discussion to vascular networks in his classic book, ‘The Fractal Geometry of Nature’.65 Vascular networks are not strictly fractal as they do not exhibit scale invariance over an infinite range of scales,66 but they show sufficient self-similarity to be treated as fractal or pseudo-fractal. The fractal dimension (Hausdorff-Besicovitch dimension) is widely used as a measure of statistical self-similarity across scaling levels and can be viewed as a measure of the effectiveness of space filling. Thus for a two-dimensional branching network (e.g. the retinal vasculature) the upper limit of the fractal dimension should be the topological dimension (i.e. 2 since a flat surface is 2 dimensional) – the closer the fractal dimension is to 2 the better the space filling. Typically the fractal dimension of healthy 2-dimensional (or 3 dimensional vascular networks analysed as a 2-dimensional slice) falls within the range 1.3–2.0.67 Fractal dimension can be viewed as complementary to older measures of vessel density68; both approaches quantify space filling, but they do not necessarily correlate closely because differences in vessel diameter affect conventional measures of density.69 Lacunarity is another measure that may be useful in describing fractal or quasi-fractal structures. Lacunarity quantifies the distribution of gap size, or lacunae, in the structure. Objects with similar fractal dimensions, may differ by lacunarity; however, as yet, lacunarity has seen only limited application to vascular networks.69–71
Takahashi72 formulated an explicit link between the fractal dimension and Murray’s law by assuming that capillaries are of uniform size and that relationship between vessel radius and length can be described by an allometric function:
Under these assumptions Takahashi shows that:
This is similar in form to Murray’s law and the branching exponent, x would be expected to be equal to D + α. Takahashi predicted that D + α should be 2.88 and observed that this was consistent with literature values for the branching exponent. Morphological studies indicate that the value of α is on average close to unity (0.76–1.21) in healthy vasculature.72 This also provides a rationale for using length to diameter ratio as a scale-independent normalization of retinal vessel structure.74 Fractal dimensions in the vasculatures range between 1.1 and 2.0,67 so one would expect x to range between ∼2 and 3 by this argument. Consistent with Takeda’s suggestion lower branch exponents were associated with lower arteriolar microvascular density in the retinal arteriolar network.75 More recently vascular networks have been treated as multifractal networks67,76 i.e. networks that show heterogeneity in scaling at different levels or regions of the network. Other approaches that have been applied to arterial networks include graph theory77 and network topological measures.78
Departures from optimal design in disease
If the normal network of small arteries is close to Murray’s optimum then it seems plausible that disease may disturb that relationship. Hutchins et al. visualised post mortem coronary arteries using an angiographic technique.79 They reported that in normal left main coronary arteries from 107 hearts the branching exponent was close to 3 (average x = 3.2) consistent with Murray’s law, but declined progressively with more severe coronary artery disease with average x = 2.2 in 17 hearts with grade 3 or 4 atherosclerosis. However the resolution of their measurements was limited to 0.1 mm and extensive characterisation of the tree could not be achieved due to the limitations of the angiographic approach. More recent work in 253 patients undergoing coronary artery intravascular ultrasound reported that deviation from Murray’s law was associated with increased coronary artery calcification.80 In pigs Zhang and Kassab reported that hypertension and LV hypertrophy was not associated with departure of coronary artery branching exponents from 3.81
The retinal circulation offers an excellent opportunity to assess branch exponents and microvascular architecture non-invasively in man.82 In a small study, Stanton et al.75 examined retinal arterioles using fluorescein angiography and reported that branch exponents were similar for normotensives (mean x = 2.7) and hypertensive individuals (mean x = 2.5), but that increased age was associated with branch exponents <3. Bifurcation angles were more acute in hypertensives (74°) than in normotensives (84°) and declined with increasing age in both groups. Chapman et al. reported that the branch exponents measured from retinal arterioles in healthy men were close to optimal (mean x = 3.1), but were significantly reduced (mean x = 2.6) in men with atherosclerosis and peripheral vascular disease.83 Witt et al. subsequently reported that increased deviation from an ‘optimal’ branch exponent of x = 3 was associated with increased future risk of coronary heart disease in an analysis of retinal photographs from the Beaver Dam Eye study.84 Another study in stroke patients also found average area ratios at branches exceeded the optimal value predicted by Murray’s Law and that more deviation from optimal was associated with ischaemic heart disease and increased periventricular white matter hyperintensities.85 Patton et al. reported that non-optimal branching of retinal arteries associated with impaired general cognitive ability and verbal fluency, whereas non-optimal branching angles were associated with reduced logical memory.86 Tillin et al. found that African-Caribbean people had less optimal retinal arteriolar branching exponents compared with Europeans87; this was suggested to be relevant to the greater risk of stroke88 and the more adverse cerebrovascular impact of high blood pressure89 in this ethnic group. Greater deviation of the branch exponent (from 3) in arteries has also been reported to be associated with proliferative retinopathy90 and peripheral neuropathy91 in adults with type 2 diabetes. Longer duration of Type 1 diabetes was also associated with an increased optimality deviation.92
Non-optimal branching geometry has also been observed in relation to adverse risk factors in early life.82 Low birth weight, a risk factor for cardiometabolic disease in adult life was associated with increased deviation of branching exponents from optimal values in children aged 11.93 Offspring of hypertensive parents (aged 9–14 y) have also been reported to have greater deviation of branch exponents from Murray’s optimal value.94
Fractal dimension has been used quite extensively to assess vascular networks in cardiometabolic and other diseases. Both reduced and increased fractal dimension has been seen in several conditions that affect the circulation (Table 1) and interpreted as indicative of a suboptimal circulatory network. Null associations have also been observed. There is some limited information regarding the relationship of fractal measures with outcomes. Low fractal dimension of the pulmonary arterial tree on CT angiography predicted poorer survival in one prospective study of people with pulmonary hypertension,95 and low fractal dimension has also been associated with an increased risk of stroke.96 Being in the higher or lower quarter of fractal dimension was associated with an increased risk of coronary heart disease mortality.97 Greater retinal fractal dimension was independently associated with early retinopathy in a study of young people (aged 12–20 y) with type 1 diabetes,98 but fractal dimension was not reported to be associated with incident retinopathy in another study of children and adolescents with type 1 diabetes.99 In older adults with type 1 diabetes a lower retinal fractal dimension was associated with complications, including proliferative retinopathy, neuropathy and older age, but not macrovascular disease.100,101
Conclusions
Application of a cost minimization approach has proved useful in the analysis of arterial networks, although it is not without limitations and a number of issues remain unresolved. Although progress has been made we need to know more about the stimuli which shape the network, how their effects are mediated, and to what extent they synergise. There is some evidence that shear stress and pressure interact,55 and a range of interactions between diverse stimuli might permit a degree of ‘weighting’ or contextualization. For instance, it has been argued the circulation might be designed to attain highest efficiency during exercise,44,102 and by implication that it is slightly suboptimal under resting conditions – effectively allowing a reserve capacity. This is consistent with some evidence, at least in terms of minimization of wave reflection.103 If this were true, the system would need to optimize its design in response to an intermittent physiological state, albeit one that may be critical for survival. How is this intermittent state sensed? One possibility might be that shear stress could have more effect on remodelling in the context of the stimuli that accompany exercise.
| Circulation | Disease/Condition | Summary of findings | Comments | References |
|---|---|---|---|---|
| Retinal | Hypertension/High BP | Lower Df | Arteries and veins not distinguished | 105–110 |
| Ischemic stroke | Lower Df | Both arteries and veins | 111 | |
| CADASIL | Lower Df | Arteries and veins not distinguished | 112 | |
| Type 1 or Type 2 Diabetes | Inconsistent | Arteries and veins not distinguished | 113–120 | |
| Cognitive dysfunction | Lower Df | Arteries and veins not distinguished | 121 | |
| Alzheimer’s disease | Lower Df | Both arteries and veins | 122 | |
| Human immunodeficiency virus (HIV) infection | Df not different | Arteries and veins not distinguished | 123 | |
| Obesity | Df not different | Arteries and veins not distinguished | 124 | |
| Glaucoma | Lower Df | Arteries and veins not distinguished | 125 | |
| Renal dysfunction/CKD | Lower Df | 126,127 | ||
| Renal | Congenitally abnormal kidneys | Lower Df | Only 2 abnormal kidneys examined | 128 |
| Pulmonary | Pulmonary Hypertension | Inconsistent | 129–131 |
Vascular fractal measures in cardiometabolic and other diseases. Abbreviations: CADASIL – Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy; CKD – chronic kidney disease; Df – fractal dimension.
Finally, is optimal design desirable? It has recently been proposed (for the bronchial tree) that an optimal network is dangerous because of its vulnerability to imperfections or vessel constriction and that a ‘safety margin’ is required in design.104 This seems difficult to reconcile with the comparative cost-insensitivity of arterial networks to small deviations from optimum, but it does raise important questions about the appropriateness of optimality as an overriding factor in circulatory design – perhaps Murray’s law should be viewed as ‘more what you’d call guidelines than actual rules.’h
Funding
AH has received financial support from the British Heart Foundation (PG/12/29/29497) and the National Institute for Health Research, University College London Hospitals Biomedical Research Centre relating to this research.
Conflicts of interest
None.
Acknowledgements
I am grateful to Nish Chaturvedi for critical reading of this paper.
Footnotes
From The Temple of Nature: Or the Origin of Society: A Poem with Philosophical Notes (1803) by Erasum Darwin. (Darwin’s notes) In branching cones, l. 259. The whole branch of an artery or vein may be considered as a cone, though each distinct division of it is a cylinder. It is probable that the amount of the areas of all the small branches from one trunk may equal that of the trunk, otherwise the velocity of the blood would be greater in some parts than in others, which probably only exists when a part is compressed or inflamed.
“Every year when the boughs of a tree have made an end of maturing their growth, they will have made, when put together, a thickness equal to that of the main stem.” Leonardo da Vinci (1515).
Keill is vague on units but Woldenberg (Woldenberg MJ. James Keill (1708) and the morphometry of the microcosm. Geometric progression laws in arterial trees. In: Stoddart DR, ed. Process and form in geomorphology. London; New York: Routledge; 1997) suggests that these are square inches.
For further information on the iatromechanists in 17th century see Brown, T. M (1970). The College of Physicians and the acceptance of iatromechanism in England, 1665–1695 Bulletin of the History of Medicine, 44(1), 12–30.
Literally translated as ‘Developmental Mechanics’; however Roux had a more causal perspective than this translation might imply.
Murray terms this factor work, but as Zamir (Zamir M. Optimality principles in arterial branching. J Theor Biol. 1976; 62:227–251) points out he is really describing power.
Taylor’s paper has been cited twice since its publication in 1967 according to Scopus (accessed 10/09/2014).
Line from Pirates of the Caribbean: The Curse of the Black Pearl (2003).
References
Cite this article
TY - JOUR AU - Alun D. Hughes PY - 2015 DA - 2015/02/24 TI - Optimality, cost minimization and the design of arterial networks JO - Artery Research SP - 1 EP - 10 VL - 10 IS - C SN - 1876-4401 UR - https://doi.org/10.1016/j.artres.2015.01.001 DO - 10.1016/j.artres.2015.01.001 ID - Hughes2015 ER -
