Point prevalence survey of antimicrobial utilization in a Canadian tertiary-care teaching hospital
- DOI
- 10.1016/j.jegh.2014.06.003How to use a DOI?
- Keywords
- Surveillance; Prospective; Acute care; Long-term care
- Abstract
Objectives: Inappropriate antimicrobial use can promote antimicrobial resistance, which is associated with increased patient morbidity and mortality. Identifying the pattern of antimicrobial use can provide data from which targeted antimicrobial stewardship interventions can be made. The primary objective was to identify the prevalence of antimicrobial use at a tertiary care teaching hospital with both acute and long-term care patients.
Methods: A point prevalence study was conducted on July 19th, 2012. Data on antimicrobial utilization, indication for prescribing, duration of therapy, and frequency of infectious disease or antimicrobial stewardship consultations were collected using a customized integrated stewardship database (SPIRIT) and prospective chart review.
Results: One or more antimicrobial agents were ordered in 31% and 4% of acute care and long-term care patients, respectively. Respiratory and urinary tract infections were the most common indication for antimicrobial therapy in both acute and long-term care. About 25% of surgical prophylaxis orders were prescribed for greater than 24 h.
Conclusion: This prospective point prevalence survey provided important baseline information on antimicrobial use within a large tertiary care teaching hospital and identified potential targets for future antimicrobial stewardship initiatives. A multi-center point prevalence survey should be considered to identify patterns of antimicrobial use in Canada and to establish the first steps toward international antimicrobial surveillance.
- Copyright
- © 2014 Ministry of Health, Saudi Arabia. Published by Elsevier Ltd.
- Open Access
- This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
1. Introduction
Antimicrobial resistance is an unavoidable consequence of antimicrobial use and is correlated with the overall use of these agents [1,2]. It has been reported that approximately one third of patients admitted to hospital receive an antimicrobial agent during their hospital stay [3–5]. This is particularly problematic when up to 50% of all courses of antimicrobial therapy are deemed unnecessary [4,6,7]. Antimicrobial resistance is increasing and is a concern because it is associated with increased patient length of hospital stay, mortality, and cost. [8,9] Since both frequent and prolonged use of antimicrobial agents may promote the emergence of resistance [10,11], antimicrobial stewardship is recommended as a means of reducing antimicrobial resistance, along with lowering the risk of adverse drug events, treatment complications, and institutional costs [7,12]. It is important for institutions to understand their patterns of antimicrobial use to identify appropriate stewardship interventions that have the greatest likelihood of impacting institutional antimicrobial utilization and therefore, the aforementioned consequences of antimicrobial use.
We conducted a point prevalence survey to quantify antimicrobial utilization and determine patterns of use at a Canadian tertiary care teaching hospital and affiliated long-term care facility. It is hoped that this information will serve as a baseline for future point-prevalence surveys, and provide a measure of the effect of antimicrobial stewardship interventions. The primary objective of this survey was to determine the prevalence of patients in the hospital and residing in the associated long-term care facility who were receiving at least one antimicrobial agent, and the prevalence of individual antimicrobial orders on the survey date. The secondary objective of this study was to describe the pattern of antimicrobial prescribing (choice of antimicrobial agent, indication, and duration of therapy).
2. Methods
The point prevalence survey was conducted at Sunnybrook Health Sciences Centre, a 1212-bed adult teaching hospital in Toronto, Ontario, Canada, with acute care facilities and an adjoining veteran’s long-term complex care facility. The hospital’s programs include aging, trauma, oncology, neurosciences, women and babies, cardiac and musculoskeletal diseases. There is a multi-disciplinary antimicrobial stewardship program (ASP), which prospectively audits the use of targeted antimicrobials and consults with prescribers on days 3, 7, and 14 of therapy [13,14]. Targeted antimicrobials include fluoroquinolones, third generation cephalosporins, carbapenems, aminoglycosides, vancomycin, and piperacillin-tazobactam. At the time of the survey, ASP services were being provided to most acute-care inpatient units but not to the long-term care units.
The survey was conducted on July 19, 2012, and included inpatients who were receiving a systemic antibacterial, antifungal, or antiviral agent on that day. Neonates, emergency room patients who were not yet admitted, palliative care patients, and obstetric patients were excluded from the survey. Orders for anti-retroviral, anti-tuberculous, and anti-parasitic medications were also excluded from the survey.
Patients were identified through the Stewardship Program Integrated Resource Information Technology (SPIRIT) database, which receives inputs in real time from the hospital’s pharmacy, microbiology, hematology and biochemistry laboratories and the admission/discharge and transfer databases [14]. The data elements of interest were abstracted directly from the SPIRIT database and supplemented by prospective review of electronic and paper charts by an infectious diseases physician or pharmacist with the aid of a standardized electronic form.
Prevalence in this study was determined using the number of patients receiving at least one antimicrobial agent or a number of antimicrobial orders as the numerator, and the total number of inpatients (excluding patients previously described) as the denominator standardized to 1000 patients on the study day. Antimicrobials in this survey were presented as classes defined by the Anatomical Therapeutic Chemical (ATC) classification system from the World Health Organization (WHO) Collaborating Centre for Drug Statistics Methodology [15]. Antimicrobial use was identified as being definitive, empiric, prophylactic, or unknown. Definitive treatment was defined as treatment that was started when either a site of infection or a pathogenic microorganism causing infection was identified. Empiric treatment was defined as treatment that was started for a presumed or possible infection without a site or infecting organism being identified. Prophylaxis was defined as the use of an antimicrobial agent in order to prevent an infection when an infection was not already present. Treatment indication was categorized as unknown if there was no identifiable reason for antimicrobial use after review of the medical records. The duration of therapy was presented as the number the days from the date of antimicrobial initiation to the date of survey.
3. Results
A total of 1021 eligible patients were identified on the survey date; 508 in acute care and 513 in long-term care. Since our long-term care facility is the largest veteran’s care facility in Canada, there was a male predominance in our long-term care resident population (53% in acute care and 86% in long term care) (Table 1).
| Description | Acute care (n = 508) | Long-term care (n = 513) |
|---|---|---|
| Age* (years) | 68 | 90 |
| 58–81 | 89–92 | |
| Male (%) | 82 (53) | 18 (86) |
| Length of stay* (days) | 7 | 245 |
| 3–18 | 35–986 |
Median and interquartile range (first quarter and third quarter) since data not normally distributed.
Demographics of patients who received antimicrobial treatment.
A total of 174 per 1000 patients were receiving at least one antimicrobial agent on the day of the study (Table 2); a prevalence of 308 per 1000 acute care patients, and 41 per 1000 long-term care residents. The majority of patients (65%) receiving antimicrobial therapy received only one antimicrobial agent (Table 2). There were a total of 244 antimicrobial orders per 1000 patients; a prevalence of 449 per 1000 acute care patients, and 41 per 1000 long-term care residents. The prevalence of patients on at least one antibiotic agent was 298 per 1000 acute care patients, and 39 per 1000 long-term care residents.
| Description | Acute care | Long-term care |
|---|---|---|
| Number of patients admitted | 508 | 513 |
| Number of patients receiving treatment (per 1000 patients) | 156 (308) | 21 (41) |
| Number of all antimicrobial orders(per 1000 patients) | 228 (449) | 21 (41) |
| Number of patients receiving one or more antimicrobial (%) | ||
| 1 | 101 (65) | 21 (100) |
| 2 | 42 (27) | 0 |
| 3 | 10 (6) | 0 |
| 4 | 2 (1) | 0 |
| 5 | 1 (0.6) | 0 |
| ⩾6 | 0 | 0 |
Prevalence of antimicrobial utilization.
In the acute setting, the majority of antimicrobial orders were for antibiotics (207/228; 91%), followed by antifungals (16/228; 7%), and antivirals (5/228; 2%). In the long-term care setting, the majority of antimicrobial orders were for antibiotics (20/21; 95%), followed by antivirals (1/21; 5%) (Fig. 1).
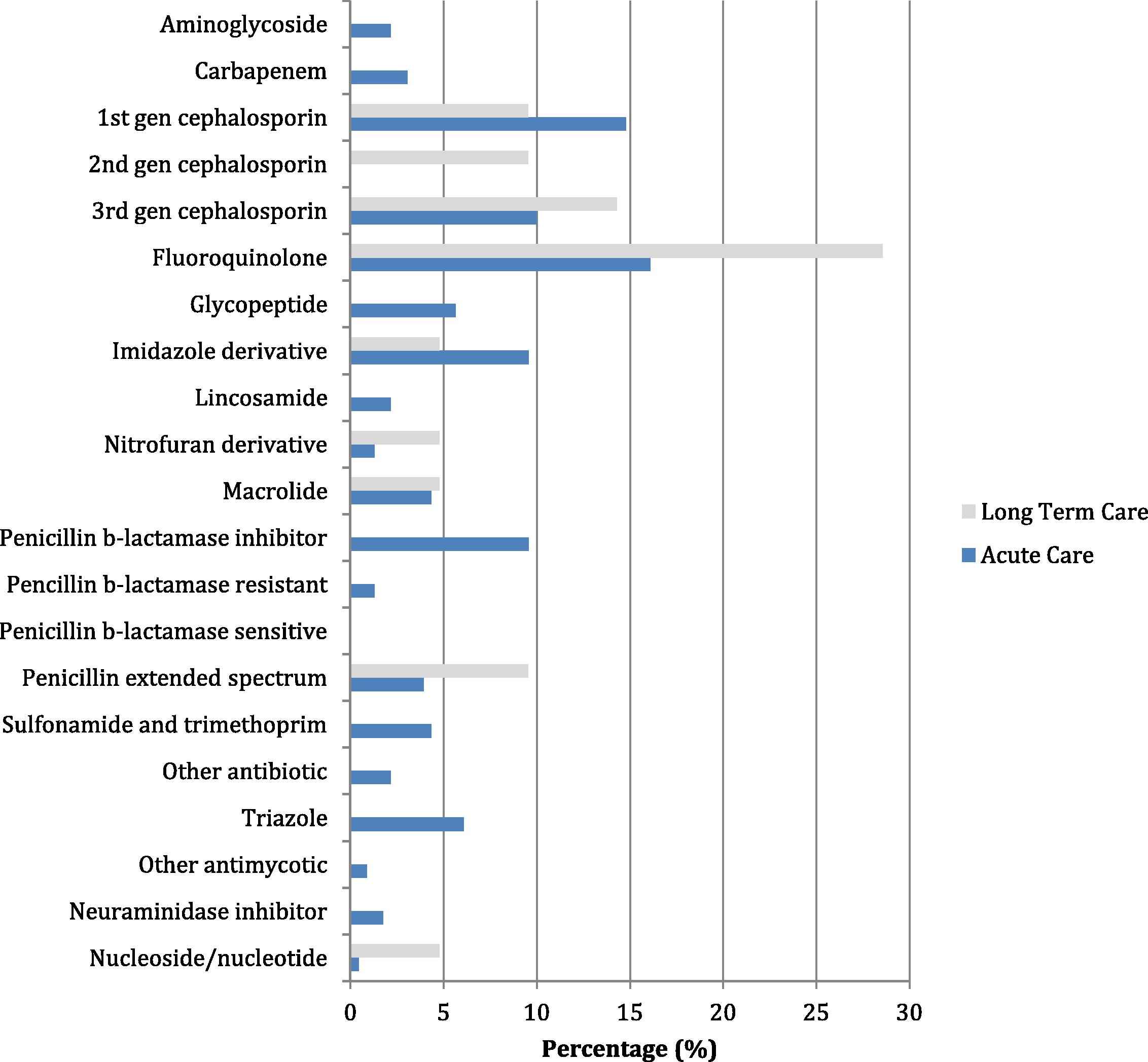
Percentage of Antimicrobial use in Acute Care and Long-term Care*.
*Denominator is the total number of antimicrobial orders in acute care or long term care. Note: aminoglycoside = tobramycin, gentamicin; carbapenem = ertapenem, meropenem; 1st cephalosporin = cefazolin, cephalexin; 2nd cephalosporin = cefuroxime; 3rd cephalosporin = ceftazidime, ceftriaxone; fluoroquinolone = ciprofloxacin, levofloxacin; glycopeptide = vancomycin; imidazole = metronidazole; lincosamide = clindamycin; macrolide = erythromycin, azithromycin, clarithromycin; penicillin b-lactamase inhibitor = amoxicillin–clavulanic acid or piperacillin–tazobactam; penicillin b-lactamase resistant = cloxicillin; penicillin b-lactamase sensitive = penicillin; penicillin extended spectrum = amoxicillin or ampicillin; other antibiotic = rifampin, trimethoprim, dapsone, or doxycycline; triazole = fluconazole, itraconazole, ketoconazole, voriconazole; other antifungal = caspofungin; nucleoside/nucleotide = acyclovir and valacyclovir.
Fluoroquinolones, cephalosporins, and penicillins were the most common classes of antibiotics ordered, accounting for more than half of all antibiotic use within the entire institution (143/227; 63%). Fluconazole was the most common antifungal prescribed, making up 50% (8/16) of all antifungal orders while oseltamivir was the most common antiviral prescribed, making up 67% (4/6) of all antiviral orders within the entire institution. Anti-pseudomonal antibiotics (aminoglycosides, carbapenems, ceftazidime, ciprofloxacin, and piperacillin–tazobactam) accounted for 30% (62/207) of antibiotics in acute care, and 10% (2/20) of antibiotics in long-term care. Ciprofloxacin accounted for 44% (27/62) of all anti-pseudomonal antibiotics used in acute care.
In the acute care setting, 38% (86/228) of all antimicrobials were ordered as an oral agent, while in long-term care, 86% (18/21) of antimicrobials were ordered as an oral agent. The use of oral formulation for antibiotics with high oral bioavailability in the acute care setting was 68% (25/37) for fluoroquinolones, 20% (1/5) for clindamycin, 27% (6/22) for metronidazole, and 67% (6/9) for co-trimoxazole. Antimicrobials were ordered most frequently for definitive treatment with highest prevalence of use in the intensive care, general medicine, and oncology units. Empiric antimicrobial therapy was most frequently used in the oncology unit (Table 3). The two most common types of infection requiring antimicrobial therapy were respiratory tract and urinary tract infections; each occurred more often in the acute care setting (Table 4).
| Medical services | Number of patients receiving an antimicrobial (Per 1000 patients to service) | Number of orders (Per 1000 patients to service) | Definite orders | Empiric orders | Unknown orders | Prophylaxis orders |
|---|---|---|---|---|---|---|
| Cardiology and cardiovascular surgery* (n = 67) | 17 (254) | 25 (374) | 18 | 3 | 0 | 4 |
| Critical care* (n = 65) | 29 (447) | 47 (724) | 37 | 4 | 1 | 7 |
| Medicine* (n = 127) | 47 (371) | 62 (489) | 55 | 6 | 0 | 2 |
| Oncology* (n = 76) | 29 (382) | 49 (645) | 34 | 15 | 3 | 1 |
| Surgery/trauma* (n = 128) | 31 (243) | 42 (329) | 29 | 2 | 3 | 8 |
Note: Orders may be counted more than once if being used for more than one indication (ie. one order may be used for a definite and empiric indication) and therefore the sum of the definite, empiric, unknown, and prophylaxis orders does not represent the total number of antimicrobials prescribed.
Total number of patients admitted to the medical service at the time of the survey.
Prevalence of antimicrobial orders and patients receiving antimicrobial treatment per service.
| Indication | Acute care (n = 508) | Per 1000 inpatients | Long-term care (n = 513) | Per 1000 inpatients |
|---|---|---|---|---|
| Definite | ||||
| Respiratorya | 43 | 85 | 11 | 22 |
| Urinary tract (symptomatic) | 24 | 48 | 1 | 2 |
| Skin and soft tissue | 17 | 32 | 4 | 6 |
| Intra-abdominal | 15 | 30 | 0 | 0 |
| Bacteremiab | 15 | 30 | 0 | 0 |
| Clostridium difficile bacterial infection | 10 | 20 | 0 | 0 |
| Bone and joint | 8 | 16 | 1 | 2 |
| Central nervous system | 4 | 8 | 0 | 0 |
| Candida (mucosal) | 3 | 6 | 0 | 0 |
| Endocarditis | 3 | 6 | 0 | 0 |
| Urinary tract (asymptomatic) | 3 | 6 | 3 | 6 |
| Dental | 2 | 4 | 0 | 0 |
| Obstetrics/gynecology | 1 | 2 | 0 | 0 |
| Other definitivec | 2 | 4 | 0 | 0 |
| Empiric | ||||
| Febrile neutropenia | 8 | 16 | 0 | 0 |
| SIRS/sepsis | 7 | 14 | 0 | 0 |
| Respiratory | 3 | 6 | 0 | 0 |
| Urinary tract | 2 | 4 | 0 | 0 |
| Intra-abdominal | 1 | 2 | 0 | 0 |
| Fever | 1 | 2 | 0 | 0 |
| Respiratory inflammation | 1 | 2 | 0 | 0 |
| Unknown | ||||
| Unknown indication | 6 | 12 | 1 | 2 |
| Prophylaxis | ||||
| Surgical | 12 | 24 | 0 | 0 |
| Medical | 7 | 14 | 0 | 0 |
Note: Patients may be counted more than once if being treated for more than one indication (ie. one patient may be concurrently treated for a urinary and respiratory tract infection) and therefore the sum of these indications does not represent the total number of patients who have received an antimicrobial agent.
Includes bacterial, viral (influenza), and fungal respiratory infections.
Includes primary, secondary, and line related bacteremia.
Given for prostate cancer and bullous pemphigoid.
Number of patients treated based on indication/site of infection.
At the time of the survey, 10% of antimicrobial orders (26/249; 1/21 in long-term care and 25/228 in acute care) had been prescribed for greater than 7 days and 6% (15/249; 1/21 in long term care and 14/228 in acute care) had been prescribed for greater than 14 days. Among the surgical prophylaxis orders, 25% (3/12) had been prescribed for greater than 24 h and 8% (1/12) had been prescribed for greater than 48 h. Of the prophylaxis orders, eleven were for cefazolin, and one was for ciprofloxacin, which was ordered by the urology service.
The ID service was consulted in the previous 7 days for 26% (6/23) of patients who received empiric therapy and 35% (43/122) of patients who received definitive antimicrobial therapy. However, ID was only consulted once for a resident in long-term care. The ASP assessed therapy within the previous 7 days for 4% (1/23) of patients who received empiric therapy and 16% (20/122) of patients who received definitive antimicrobial therapy; all ASP assessments occurred in acute care.
4. Discussion
Point prevalence surveys have been utilized extensively to examine antimicrobial use in a number of European countries [16–22]. To our knowledge, this current study represents one of the first point prevalence surveys designed to investigate antimicrobial use among hospitalized patients in a Canadian adult tertiary care center. In the acute care setting, nearly one third of hospitalized patients were on at least one antimicrobial agent, and the prevalence of antimicrobial orders was 449 per 1000 patients. These findings were similar to that reported in a large multi-national European point prevalence survey (2009 ESAC) where 29% of patients received an antimicrobial agent and the prevalence of antimicrobial orders was 407 per 1000 patients [18]. These findings were also consistent with other smaller point prevalence surveys conducted in a number of individual European countries [16,17,20,21].
Similar to other point-prevalence surveys, β-lactams were the most commonly used class of antibiotics in the acute care setting. However, when comparing antibiotics within the β-lactam class, our survey showed an overall lower use of penicillins and higher use of cephalosporins compared to other surveys [16–18,23,24]. This difference was mostly driven by the greater use of first generation cephalosporins and lesser use of penicillins with β-lactamase inhibitors. Furthermore, the prevalence of fluoroquinolone use in the acute care setting in our survey (16%) was notably higher and almost double than that reported in the 2009 ESAC survey (9%) [18]. Interestingly, another Canadian point prevalence survey, although primarily designed to investigate the prevalence of hospital acquired infections, showed a similar use of penicillins, cephalosporins, and fluoroquinolones as in our study [25]. In contrast to acute care, the prevalence of patients on antimicrobial therapy and the prevalence of antimicrobial orders in the long-term care setting were both lower. Although β-lactams and fluoroquinolones were also most commonly prescribed in the long-term care setting, the proportion of residents treated with a third generation cephalosporin or fluoroquinolone were higher compared to patients in acute care. More than 25% of residents on antibiotics in long-term care were treated with a fluoroquinolone. Our results were similar to those found in a Canadian point prevalence survey among long-term care facilities in Ontario, with the exception that third generation cephalosporin use was markedly higher in our survey (14% vs. 1.2%) [26]. The reason for this difference may be due to increased accessibility to intravenous medications at our hospital, since it services both acute and long-term care patients concurrently. A point prevalence survey of European nursing homes also showed a similar prevalence of overall antimicrobial use, again with a higher use of penicillin antibiotics, particularly amoxicillin–clavulanic acid (accounting for 40% of all penicillin use) [19]. The reason for differences in antimicrobial prescribing habits between countries is complex, and has been reported in a previous study comparing antimicrobial consumption between North America and Europe [27]. Although difficult to fully elucidate, differences may be due to different opinions and traditions regarding infection treatment, differences in antimicrobial resistance and guideline adherence, aggressive marketing of newer agents, and differences in antibiotic-control programs [27,28]. Other clinicians propose that the type of health care financing systems, number of doctors per inhabitant, and the average time spent with patients may also lead to different prescribing habits [29]. Also, unlike in Europe, there are no ongoing regional initiatives in North America to capture data on antimicrobial use, which may have downstream impact on overall antimicrobial consumption if such a surveillance system was available. The results are also difficult to compare since most point prevalence surveys do not provide an assessment of the appropriateness of therapy, which may also impact on the overall prevalence of antimicrobial use.
Respiratory tract and urinary tract infections were the most common indications for definitive therapy in either acute or long-term care patients. These were also the predominant indication for fluoroquinolone use in our survey. Therefore, antimicrobial stewardship initiatives targeting these indications may have the greatest potential to impact on patient care, antimicrobial use, incidence of nosocomial infections (i.e. Clostridium difficile), and resistance [13,30,31]. Another area for antimicrobial stewardship intervention, as suggested in previous surveys [16,18], is with antimicrobial surgical prophylaxis. Among patients receiving surgical prophylaxis, approximately one-quarter of patients were receiving prophylaxis for greater than 24 h at the time of this survey. The majority of these cases would not have been eligible for ASP review in our facility because of the use of a non-targeted antibiotic, and may present as an opportunity for further improvement of the ASP at our institution.
Finally, there were fewer ID consults for long-term care residents, likely due to lower severity and complexity of infections in residents who could continue to receive treatment for their infections in the long-term care facility without being transferred to acute care. The overall lower frequency of ASP consults compared to ID consults was expected because the ASP only consulted on patients who received targeted antimicrobials on selected days of antimicrobial therapy in acute care. At the time of this survey, ASP services were not available to long-term care residents. Given the prevalence of broad spectrum antibiotic use and the indications for antimicrobial use found in our survey, as of September 16, 2013, we instituted an ASP initiative in our long-term care facility.
This study has important limitations inherent to the use of point prevalence surveys. Point prevalence surveys look at a single point in time and so the results can be affected by normal day-to-day variation, existing trends, and seasonality of antimicrobial use. Different antimicrobial agents can be chosen for identical indications depending on the prescriber’s judgment, which was not captured in this study. Point prevalence surveys may also over-represent antibiotic treatments that are given for longer durations since they are more likely to overlap the study date. Although highly trained infectious diseases clinicians conducted the data collection using a standardized form, there still may be some differences in the interpretation of data from incomplete charts, potentially leading to discrepancies in assessments. Also, this survey was conducted at a single university-affiliated health center with policies and programs in place to optimize antimicrobial prescribing. So, differences in patient population and prescribing practices should be considered when comparing these results to other institutional point prevalence surveys. But given the availability of national treatment guidelines and similarities in prescribing practices among academic teaching hospitals, the results of this survey provides a good initial estimation of institutional antimicrobial use in Canada. Unfortunately, given the limited resources and infrastructure, a Canadian multi-center point prevalence survey will not likely be initiated in the near future. Finally, there is no international standardized hospital categorization system, which makes comparing hospitals from different countries difficult.
5. Conclusion
This study provides important insight into the use of antimicrobial agents and prescribing patterns at a Canadian tertiary care center. These results will provide direction to future endeavors to further optimize the use of antimicrobials at our institution. Moreover, the results of this survey brings to light the differences in antimicrobial use between countries, and highlights the need for a standardized nation-wide Canadian point-prevalence survey to better understand how antimicrobials are being used in Canada and to establish the first steps toward international antimicrobial surveillance.
Funding
No financial support was received by any of the authors for conducting this survey. All data have been generated as part of the routine work of this institution.
Transparency declaration
All authors: No reported conflicts to declare
Acknowledgement
Results have been previously presented as a poster at the 2013 Association of Medical Microbiology and Infectious Disease (AMMI) Canada conference.
References
Cite this article
TY - JOUR AU - Colin Lee AU - Sandra A.N. Walker AU - Nick Daneman AU - Marion Elligsen AU - Lesley Palmay AU - Bryan Coburn AU - Andrew Simor PY - 2014 DA - 2014/08/08 TI - Point prevalence survey of antimicrobial utilization in a Canadian tertiary-care teaching hospital JO - Journal of Epidemiology and Global Health SP - 143 EP - 150 VL - 5 IS - 2 SN - 2210-6014 UR - https://doi.org/10.1016/j.jegh.2014.06.003 DO - 10.1016/j.jegh.2014.06.003 ID - Lee2014 ER -
